Correct option is B
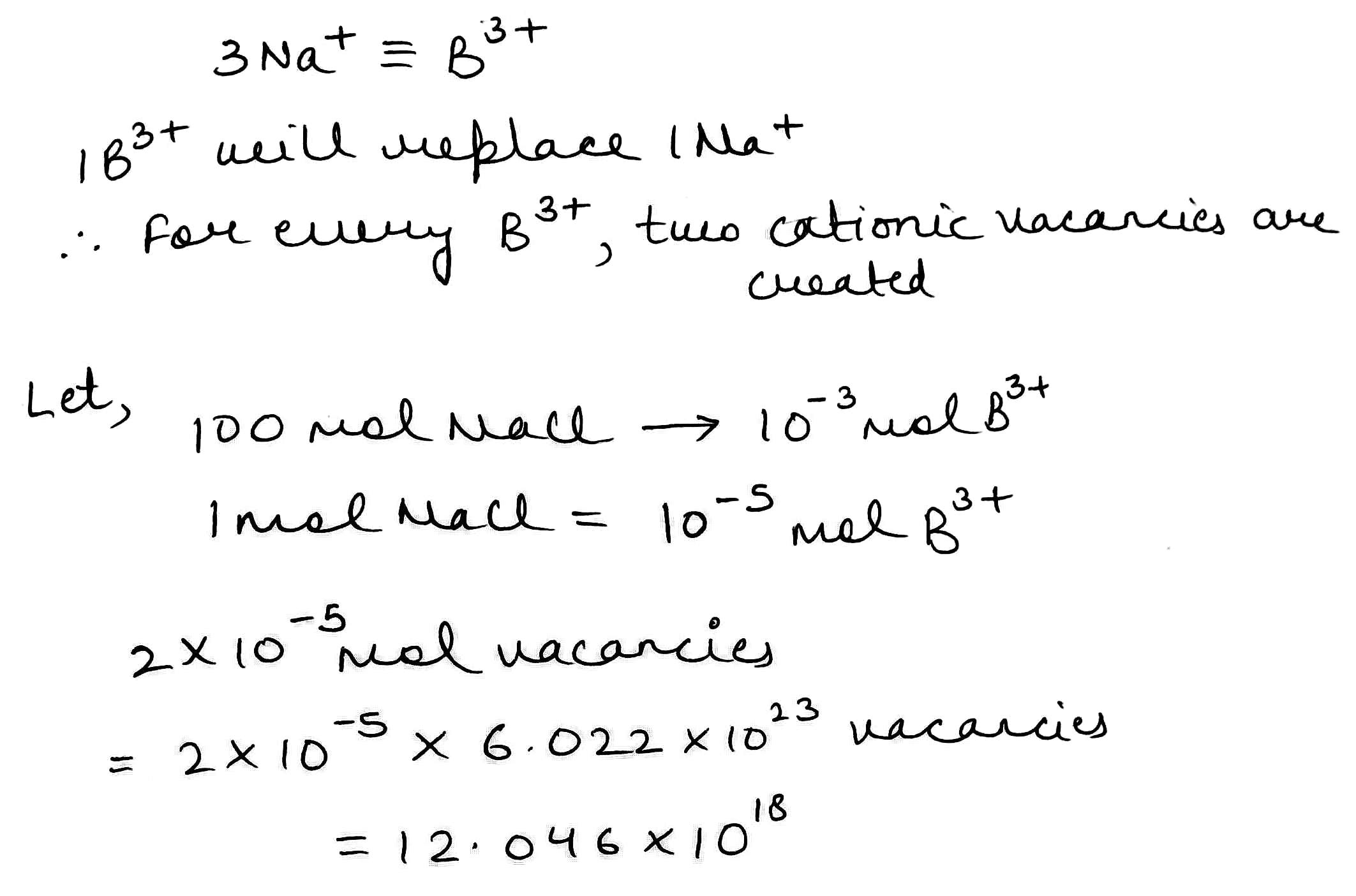
Due to the addition of BCl3, each B3+ion replaces three Na+ ions, but occupies one Na+ lattice point. Thus, this exchange of Na+ ion by B3+ ion makes two cationic vacancies.
The number of cation vacancies per mole,
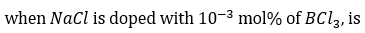
Due to the addition of BCl3, each B3+ion replaces three Na+ ions, but occupies one Na+ lattice point. Thus, this exchange of Na+ ion by B3+ ion makes two cationic vacancies.
The system with the largest internal pressure is
A compound consists of atoms A and B. Atoms of B form cubic close packing lattice and atoms of A occupy half of octahedral voids and half of tetrahedral voids. What is the molecular formula of the compound?
Entropy associated with a die with all its six faces having equal probability of being on top is (kB : Boltzmann constant):

The equivalent symmetry operations for and are, respectively,
The compound that will show the following characteristic splitting pattern in the 1H NMR spectrum is

For CO, the contour plot of the molecular orbital which best represents the HOMO is
The degree of polymerization at t = 10h of a polymer formed by a stepwise process with polymerization rate constant of and an initial monomer concentration of 50 mM is
Iron belongs to the BCC lattice. The Miller indices of the second allowed reflection in the powder diffraction pattern of iron would be
A gas is known to satisfy Langmuir isotherm when adsorbed on a certain metal surface. If the fractional coverage of the gas is 0.5 when the gas pressure is 1.0 Pa, the fractional coverage at 3.0 Pa would be closest to

