Correct option is B
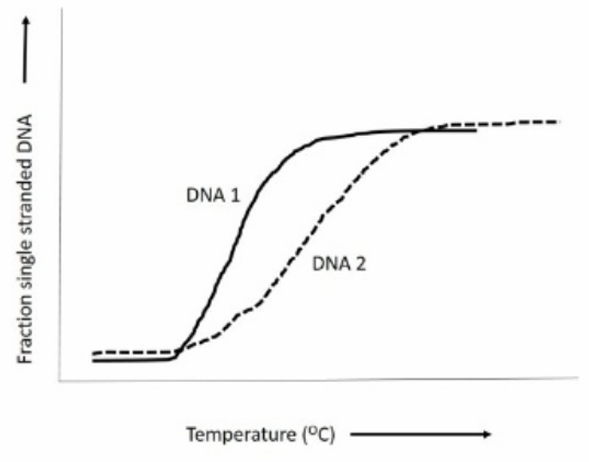
The melting curve shows the fraction of single-stranded DNA as the temperature increases. The higher the GC content, the more stable the DNA molecule is, which means it will require a higher temperature (Tm) to denature.
DNA1 reaches its transition point (where the melting begins) at a lower temperature compared to DNA2. Since GC base pairs are more stable due to three hydrogen bonds (compared to two hydrogen bonds in AT pairs), a DNA molecule with higher GC content will have a higher Tm.
DNA2, which melts at a higher temperature, must therefore have more GC content than DNA1. This indicates that DNA1 has a lower GC content than DNA2, which makes option (2) correct.
Information Booster:
GC content is directly correlated with the stability of DNA, as GC pairs form three hydrogen bonds, making them more resistant to melting than AT pairs, which form two hydrogen bonds.
The Tm (melting temperature) of DNA increases with the GC content because GC pairs are more stable and harder to separate than AT pairs.
In the provided melting curve, DNA1 begins melting at a lower temperature, suggesting it has fewer GC pairs, hence a lower GC content than DNA2.
AT content is inversely related to GC content; as GC content increases, AT content decreases.
Additional Knowledge:
Option 1: DNA1 has a lower AT content than DNA2 – This is incorrect because the higher melting temperature of DNA2 suggests it has a higher GC content, which means DNA1 has a higher AT content than DNA2.
Option 3: The DNA1 solution has a sequence-independent dsDNA binding protein – The question does not provide any information about proteins or their binding, so this option is not relevant.
Option 4: DNA2 has a high number of mismatched nucleotides – The melting curve does not provide information on mismatched nucleotides, so this option is not supported by the data.