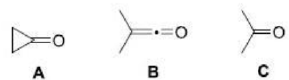
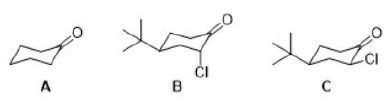
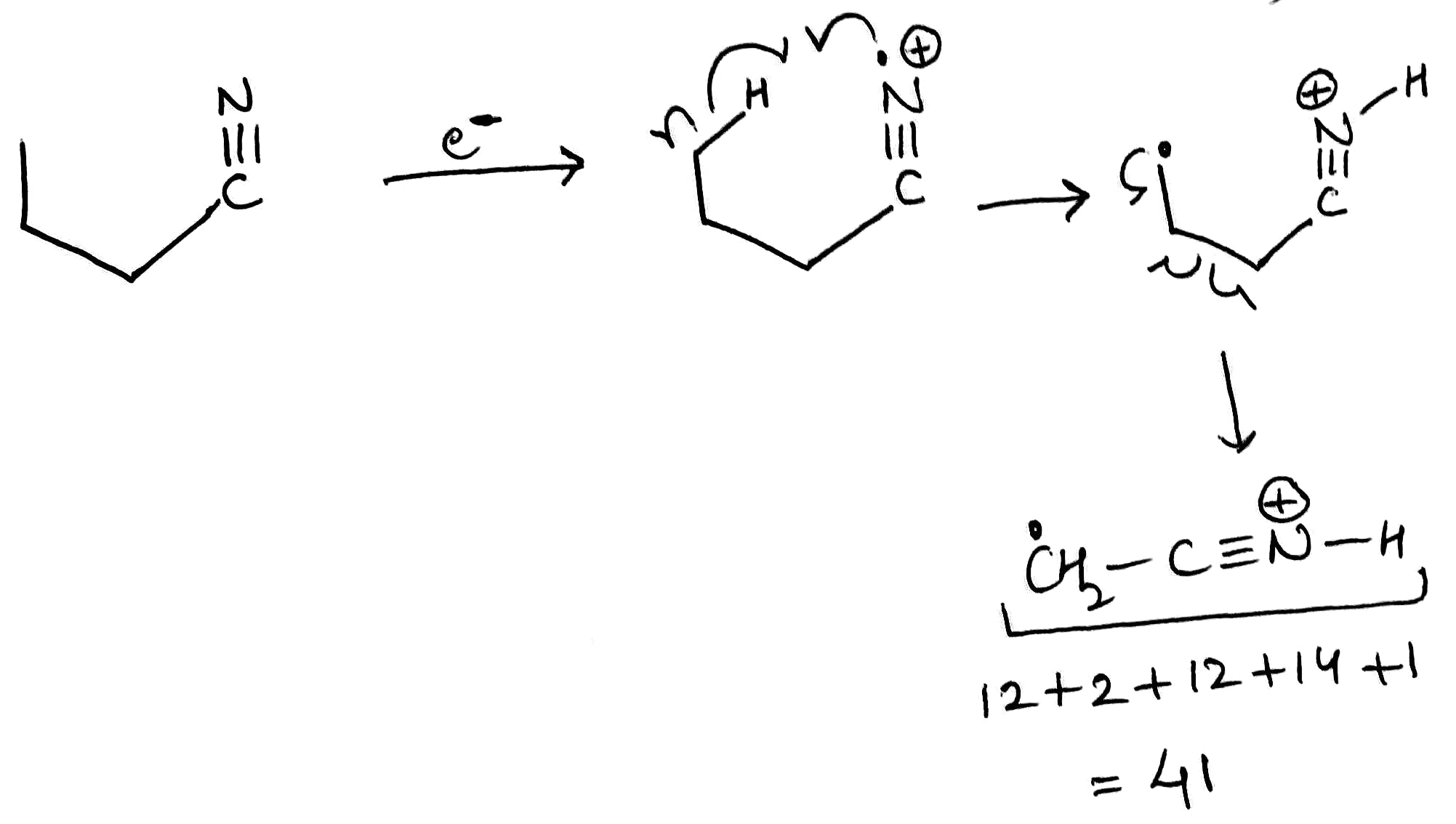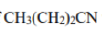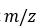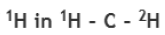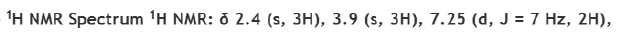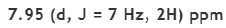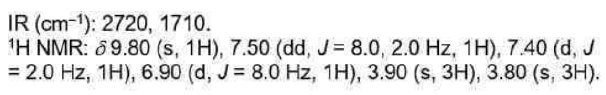Correct option is C
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. In a typical MS procedure, a sample, which may be solid, liquid, or gaseous, is ionized, for example by bombarding it with a beam of electrons. This may cause some of the sample's molecules to break up into positively charged fragments or simply become positively charged without fragmenting. These ions (fragments) are then separated according to their mass-to-charge ratio.
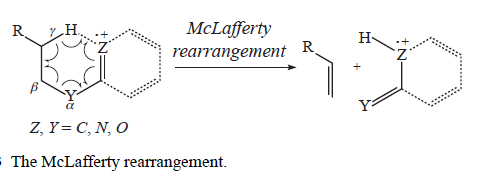
A very common fragmentation that can occur with many substrates is the McLafferty rearrangement. In the McLafferty rearrangement, a hydrogen atom on a carbon 3 atom away from the radical cation of an alkene, arene, carbonyl, or imine (a so-called γ-hydrogen) is transferred to the charge site via a six-membered transition state, with concurrent cleavage of the sigma bond between the α and β positions of the tether. This forms a new radical cation and an alkene with a π bond between what were the original β and γ carbons. For simplicity, the mechanism of the McLafferty rearrangement is usually drawn as a concerted process. The McLafferty rearrangement is readily observed in the mass spectra of many organic functional groups.