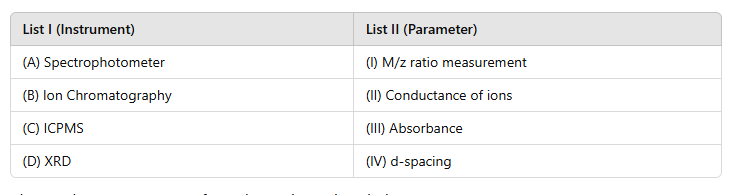
Correct option is A
Introduction:
- Chromatography is a powerful set of laboratory techniques used for the separation of mixtures. The technique relies on the differential distribution of the components of a mixture between two phases: a stationary phase and a mobile phase.
- The components travel at different speeds through the stationary phase, driven by the mobile phase, leading to their separation.
- The nature of the stationary phase is the defining characteristic that determines the type of interaction (e.g., adsorption, partition, ion exchange) and the specific chromatographic method employed.
Information Booster:
- Paper Chromatography: This is a form of Partition Chromatography. The cellulose of the paper sheet acts as an inert solid support.
- The actual stationary phase is the water that is naturally absorbed (held) within the cellulose fibers. The separation occurs because the components of the mixture partition (distribute) between the liquid mobile phase and the stationary liquid (absorbed water).
- Thin Layer Chromatography (TLC) : This is typically a form of Adsorption Chromatography. The stationary phase is a thin layer of fine adsorbent material, usually silica gel ($SiO_2$) or alumina ($Al_2O_3$), which is coated on an inert, flat backing (like glass or plastic).
- Separation occurs as components selectively adsorb (stick) and desorb from the surface of the fine particles.
- High Performance Liquid Chromatography (HPLC) : This is a highly advanced column technique that can use various separation mechanisms.
- The stationary phase consists of very small, spherical particles (like porous silica) with a very high surface area. These are densely packed into a rigid column.
- The small particle size is what necessitates the high pressure (or "High Performance") to force the liquid mobile phase through the column, providing excellent separation efficiency.
- Gas Liquid Chromatography (GLC) : In this method, the mobile phase is an inert gas (e.g., helium).
- The stationary phase is a non-volatile liquid (e.g., a high-boiling point polymer) that is coated onto a solid support material and packed into a column (or coated directly on the inside wall of a capillary column).
- The column is kept in an oven to ensure the sample components remain in the vapor state. Separation is based on the differential partition of the volatile components between the mobile gas phase and the stationary liquid phase.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins