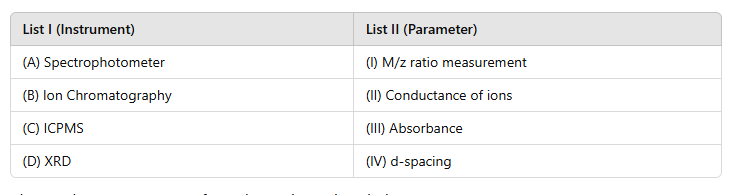
Correct option is C
The correct answer is
(c) Adsorption chromatography.
Thin Layer Chromatography (TLC) is a form of
adsorption chromatography where the separation of compounds is based on their differing affinities for the stationary phase and the mobile phase. In TLC:
1.
Stationary Phase: A thin layer of adsorbent, such as silica gel or alumina, coated on a glass, plastic, or metal plate.
2.
Mobile Phase: A liquid solvent or mixture of solvents that moves through the stationary phase by capillary action.
3.
Adsorption Mechanism: The analyte molecules adhere to the surface of the stationary phase. The extent of adsorption determines how far each component travels up the plate.
Compounds with higher affinity for the stationary phase travel slower, while those with lower affinity travel faster, enabling separation.
Information Booster:
1.
Adsorption Chromatography:
· Relies on the adhesion of solute molecules to a solid surface.
· Used for separating non-volatile mixtures.
· Examples:
TLC and
Column Chromatography.
2.
Stationary Phase:
· Commonly used adsorbents:
Silica gel, Alumina, and Cellulose.
3.
Applications of TLC:
·
Qualitative analysis of compounds.
·
Purity testing.
· Separation of
plant extracts, amino acids, and drugs.
4.
Rf Value:
· The
Retention Factor (Rf) is calculated as:
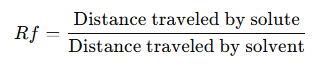
· Helps in identifying compounds based on their movement.
Additional Knowledge:
1.
(a) Partition Chromatography:
· Based on partitioning between two immiscible phases (liquid-liquid).
· Example:
Paper Chromatography.
2.
(b) Ion-exchange Chromatography:
· Separation of ions based on their affinity for ion-exchange resins.
· Common in protein purification.
3.
(c) Adsorption Chromatography:
· Involves adsorption of solutes on a solid stationary phase (e.g., TLC).
4.
(d) Size-exclusion Chromatography:
· Separates molecules by size using a porous stationary phase.
· Used for
proteins and polymers.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins