Correct option is D
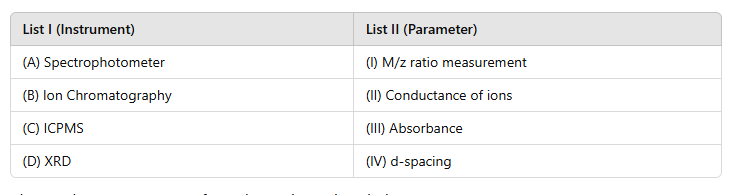
The correct answer is (d) A-III, B-II, C-I, D-IV. The instruments and their corresponding parameters are:
1. (A) Spectrophotometer → (III) Absorbance
· A spectrophotometer measures the absorbance of light by a sample at specific wavelengths.
2. (B) Ion Chromatography → (II) Conductance of ions
· Ion chromatography is used to separate and measure ions in a sample based on their conductance.
3. (C) ICPMS (Inductively Coupled Plasma Mass Spectrometry) → (I) M/z ratio measurement
· ICPMS measures the mass-to-charge (M/z) ratio of ions, allowing for elemental and isotopic analysis.
4. (D) XRD (X-Ray Diffraction) → (IV) d-spacing
· XRD measures d-spacing, which is the distance between atomic planes in a crystal structure.
Information Booster: 1. Spectrophotometer:
· Used in chemistry and biology to measure how much light a sample absorbs.
2. Ion Chromatography:
· An analytical method to separate and detect ions in a solution.
3. ICPMS (Inductively Coupled Plasma Mass Spectrometry):
· A sensitive technique for detecting metals and isotopes by measuring their M/z ratios.
4. XRD (X-Ray Diffraction):
· Used for determining crystal structures by analyzing diffraction patterns.
Additional Knowledge: · Absorbance (A-III): Quantified using the Beer-Lambert Law.
· Conductance of Ions (B-II): Key for analyzing water quality and ionic composition.
· M/z Ratio (C-I): Fundamental for identifying elements and isotopes in mass spectrometry.
· d-Spacing (D-IV): Essential for identifying crystalline phases in materials science.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins

