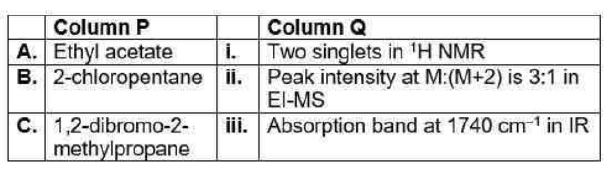
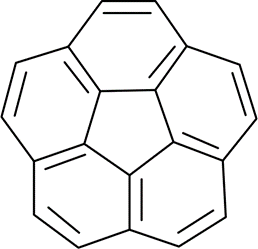
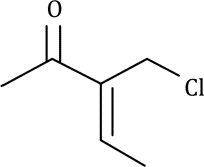
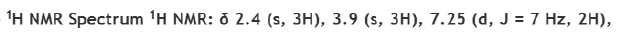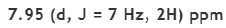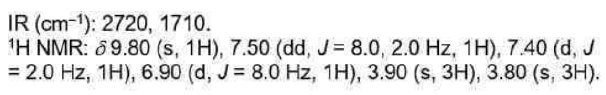Correct option is A
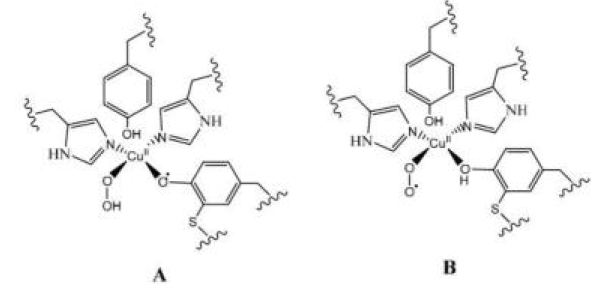
Room Temperature EPR Spectroscopy: Electron Paramagnetic Resonance (EPR) spectroscopy is a technique used to study materials with unpaired electrons, such as radicals or metal complexes. It is sensitive to the magnetic environment of electrons. Since A and B do not have significant differences in their unpaired electron environment, this method is not useful for distinguishing them.
Vibrational Spectroscopy: This method, which includes techniques such as IR and Raman spectroscopy, studies the vibrational modes of molecules. Different functional groups within molecules absorb at characteristic frequencies, allowing for the distinction between molecular structures. This method can effectively distinguish compounds A and B as their structures differ, leading to different vibrational frequencies, especially in regions associated with their functional groups.
Electrospray lonisation Mass Spectrometry (ESI-MS): ESI-MS is a mass spectrometric technique that ionizes large biomolecules into the gas phase to measure their mass-to-charge ratio. It provides information about the molecular weight and structure. It is most likely that A and B have the same molecular weights (as they are intermediates in the same pathway), this technique may not effectively differentiate between them.
Electronic Spectroscopy: This technique measures the absorption or emission of light by molecules as electrons transition between different energy levels. Changes in the electronic structure of a compound can be observed through its spectral signature. Differences in the electronic structure of A and B can be detected through this method, as electronic transitions (e.g., absorption peaks) will vary between the two compounds. Thus, electronic spectroscopy can effectively distinguish between A and B.