Correct option is A
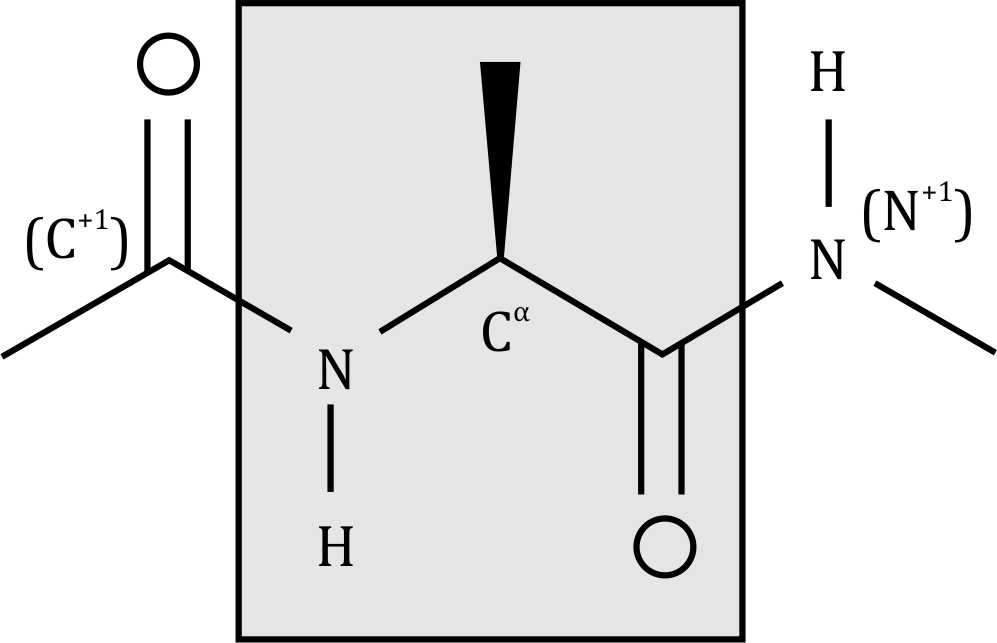
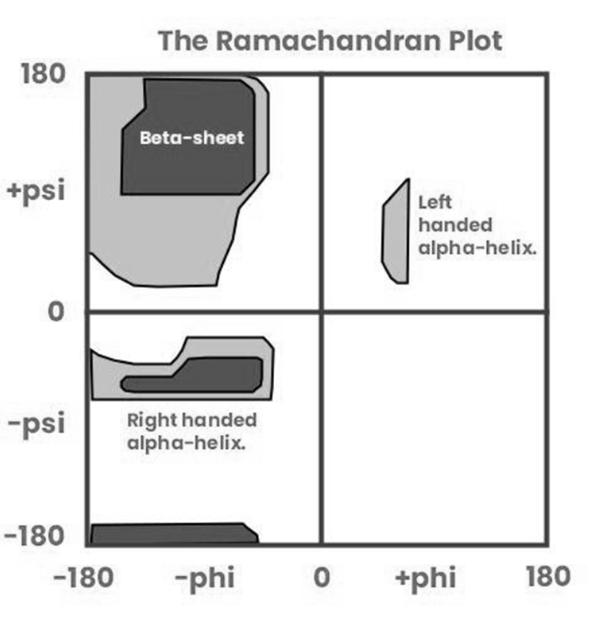
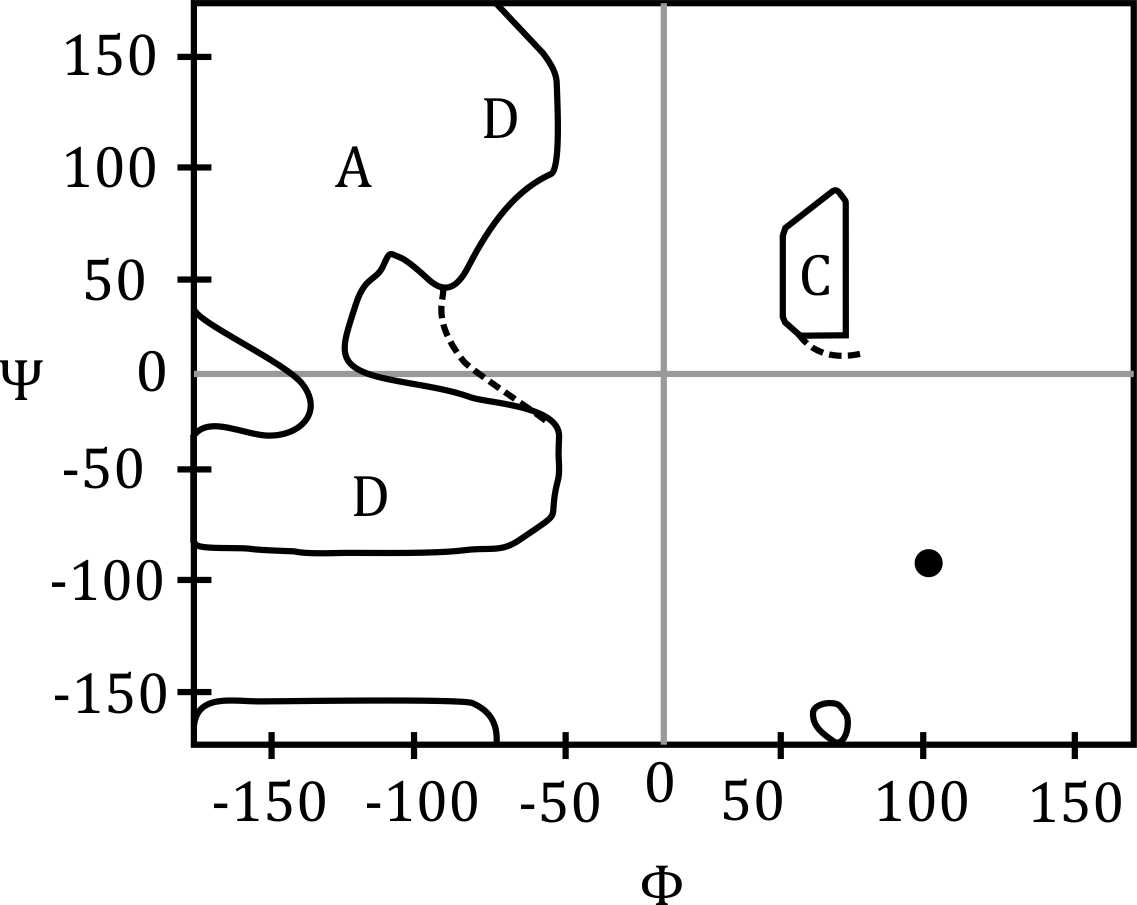
The Ramachandran plot is used to study the conformational angles of polypeptide chains. The torsion angles ϕ (phi) and ψ (psi) are defined by the backbone atoms of the peptide bond. These angles are crucial for determining the secondary structure of proteins.
1. Definition of ϕ (phi) angle:
- The ϕ angle describes the rotation around the N-Cα bond.
- It is measured using four atoms: C⁻¹, N, Cα, and C.
- C⁻¹ (carbonyl carbon of the previous amino acid)
- N (amide nitrogen of the current residue)
- Cα (alpha carbon of the current residue)
- C (carbonyl carbon of the current residue)
2. Definition of ψ (psi) angle:
- The ψ angle describes the rotation around the Cα-C bond.
- It is measured using four atoms: N, Cα, C, and N⁺¹.
- N (amide nitrogen of the current residue)
- Cα (alpha carbon of the current residue)
- C (carbonyl carbon of the current residue)
- N⁺¹ (amide nitrogen of the next residue)
Information Booster:
- Peptide bonds are planar due to partial double-bond character, restricting rotation.
- ϕ and ψ angles determine secondary structures like α-helices and β-sheets.
- Glycine has a larger allowable Ramachandran space due to its small side chain (H).
- Proline restricts ϕ rotation because of its cyclic structure.
- Trans conformation is more stable than cis for peptide bonds (except in proline).
- Steric hindrance affects the allowed regions of the Ramachandran plot.
- Proteins fold into stable conformations by optimizing ϕ and ψ angles.