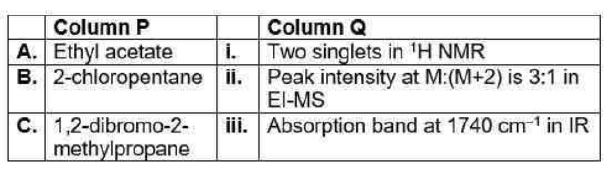
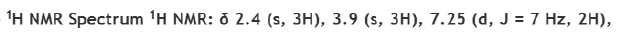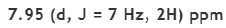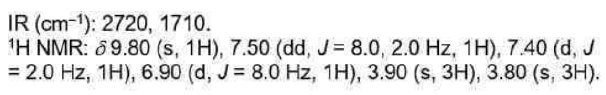Correct option is D
Proton-Decoupled 13C NMR provides a spectrum where signals from 13C nuclei appear without splitting due to attached protons. This simplifies the spectrum and aids in identifying unique carbon environments. In proton-decoupled 13C NMR, the signal intensities vary for several reasons.
Nuclear Overhauser Effect (NOE): When we obtain a proton-decoupled 13C spectrum, the intensities of many of the carbon resonances increase significantly above those observed in a proton-coupled experiment. Carbon atoms with hydrogen atoms directly attached are enhanced the most, and the enhancement increases (but not always linearly) as more hydrogens are attached. This effect is known as the Nuclear Overhauser effect, and the degree of increase in the signal is called the Nuclear Overhauser enhancement (NOE).
Additionally, different carbons have varying relaxation times (T1), impacting the time it takes for nuclei to return to equilibrium after excitation. These factors cause inconsistencies in the observed signal intensity, making integration unreliable for quantifying carbon atoms.
Poor isotopic abundance of 13C (1.1%) affects the overall signal strength but does not affect relative intensities in the same way as NOE and relaxation differences do.
Information Booster:
The Nuclear Overhauser effect is general, showing up when one of two different types of atoms is irradiated while the NMR spectrum of the other type is determined. If the absorption intensities of the observed (i.e., nonirradiated) atom change, enhancement has occurred. The effect can be either positive or negative, depending on which atom types are involved. In the case of 13C interacting with 1H, the effect is positive; irradiating the hydrogens increases the intensities of the carbon signals. Relaxation involves two main processes: longitudinal (T1) and transverse (T2). Different carbons relax at different rates depending on their environment, bond structures, and local dynamics, affecting signal intensities.