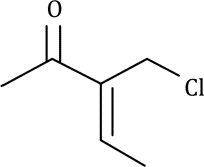
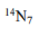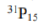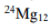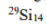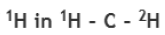Correct option is C
The key determinant of NMR activity in atomic nuclei is the nuclear spin quantum number (I). This intrinsic quantum property, similar to an atom's "spin", characterizes the angular momentum of the nucleus. To be NMR-active, a nucleus must have a non-zero nuclear spin (I≠ 0).It is this non-zero spin that enables nuclei to interact with external magnetic fields and show signals in NMR. Atoms with an odd sum of protons and neutrons exhibit half-integer values for the nuclear spin quantum number (I= 1/2, 3/2, 5/2, and so on). These atoms are NMR-active because they possess non-zero nuclear spin. Atoms with an even sum but both an odd number of protons and an odd number of neutrons exhibit integer nuclear spins (I= 1, 2, 3, and so on). Conversely, atoms with an even number of both protons and neutrons have a nuclear spin quantum number of zero (I= 0), and therefore are not NMR-active. NMR-active nuclei, particularly those with a spin quantum number of 1/2, are of great significance in NMR spectroscopy. Examples include1H,13C,15N, and31P. Some atoms with very high spin (as 9/2 for99Tc atom) are also extensively studied with NMR spectroscopy.
In option C, mass number is 24 and atomic number is 12. Therefore, number of neutrons=24-12=12. As both the number of protons and neutrons are even, the nuclei is NMR inactive.