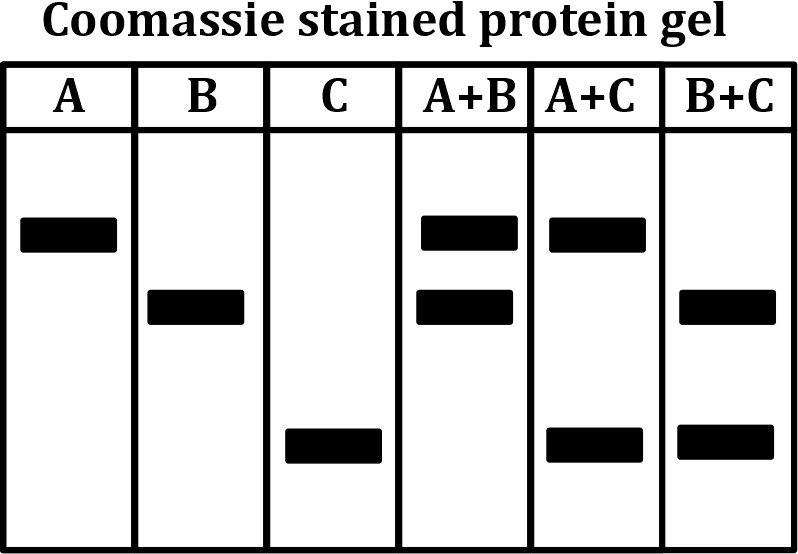
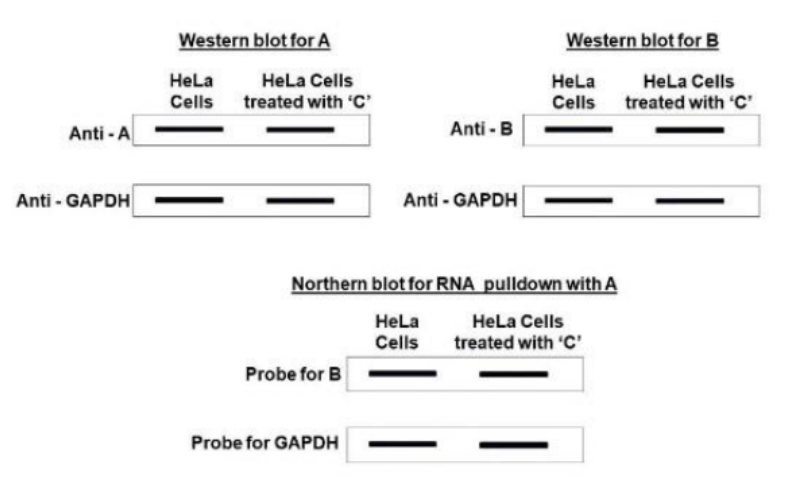
Correct option is B
A pulse oximeter works by shining light through the fingertip (or earlobe) and measuring the absorption of light at two specific wavelengths. These wavelengths are chosen because oxyhemoglobin (hemoglobin bound with oxygen) and deoxyhemoglobin (hemoglobin without oxygen) absorb light at different rates in specific regions of the light spectrum.
The two main wavelengths used by pulse oximeters are:
- Visible light (~660 nm): This wavelength is absorbed more by deoxyhemoglobin (hemoglobin without oxygen).
- Infrared light (~940 nm): This wavelength is absorbed more by oxyhemoglobin (hemoglobin bound with oxygen).
By comparing the differential absorption of these two wavelengths, the pulse oximeter can calculate the percentage of oxygen saturation in the blood (SpO2).
Information Booster:
- Oxyhemoglobin absorbs more infrared light than deoxyhemoglobin, while deoxyhemoglobin absorbs more visible red light than oxyhemoglobin.
- The difference in absorption between these two wavelengths is used to calculate blood oxygen saturation.
- Pulse oximeters are designed to use non-invasive optical sensors, allowing for continuous monitoring of oxygen levels in patients.
- Visible light typically has wavelengths between 400 to 700 nm, while infrared light ranges from 700 nm to 1,500 nm.
- The pulsating nature of the signal helps differentiate the absorption from the static tissue and bone, ensuring that the measurement is taken from the blood.