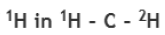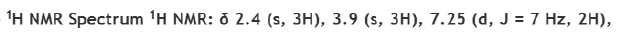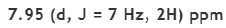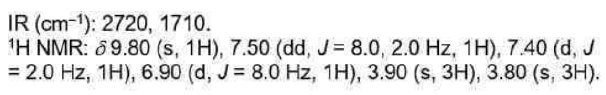Correct option is D
Nuclear magnetic resonance spectroscopy, most commonly known as NMR spectroscopy, is a spectroscopic technique based on re-orientation of atomic nuclei with non-zero nuclear spins (Examples include 1H, 13C, 15N, and 31P) in an external magnetic field. This re-orientation occurs with absorption of electromagnetic radiation in the radio frequency region from roughly 4 to 900 MHz, which depends on the isotopic nature of the nucleus and increased proportionally to the strength of the external magnetic field.
Nuclear magnetic resonance has great utility because not all protons in a molecule have resonance at exactly the same frequency. This variability is due to the fact that the protons in a molecule are surrounded by electrons and exist in slightly different electronic (magnetic) environments from one another. The valence-shell electron densities vary from one proton to another. The protons are shielded by the electrons that surround them. In an applied magnetic field, the valence electrons of the protons are caused to circulate. This circulation, called a local diamagnetic current, generates a counter magnetic field that opposes the applied magnetic field. Figure illustrates this effect, which is called diamagnetic shielding or diamagnetic anisotropy.
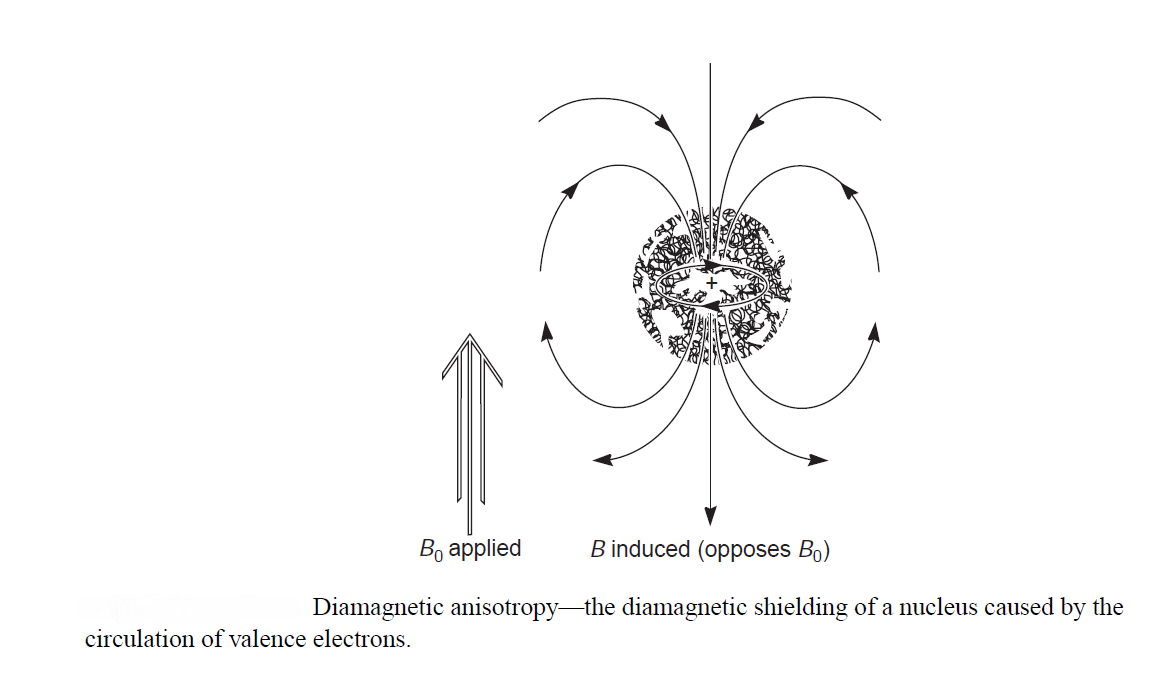
Circulation of electrons around a nucleus can be viewed as being similar to the flow of an electric current in an electric wire. From physics, we know that the flow of a current through a wire induces a magnetic field. In an atom, the local diamagnetic current generates a secondary, induced magnetic field that has a direction opposite that of the applied magnetic field.
As a result of diamagnetic anisotropy, each proton in a molecule is shielded from the applied magnetic field to an extent that depends on the electron density surrounding it. The greater the electron density around a nucleus, the greater the induced counter field that opposes the applied field. The counter field that shields a nucleus diminishes the net applied magnetic field that the nucleus experiences. As a result, the nucleus precesses (the precession of the magnetic moment of an object about an external magnetic field) at a lower frequency. This means that it also absorbs radiofrequency radiation at this lower frequency. Each proton in a molecule is in a slightly different chemical environment and consequently has a slightly different amount of electronic shielding, which results in a slightly different resonance frequency.
These differences in resonance frequency are very small. For instance, the difference between the resonance frequencies of the protons in chloromethane and those in fluoromethane is only 72 Hz when the applied field is 1.41 Tesla. Since the radiation used to induce proton spin transitions at that magnetic field strength is of a frequency near 60 MHz, the difference between chloromethane and fluoromethane represents a change in frequency of only slightly more than one part per million! It is very difficult to measure exact frequencies to that precision; hence, no attempt is made to measure the exact resonance frequency of any proton. Instead, a reference compound is placed in the solution of the substance to be measured, and the resonance frequency of each proton in the sample is measured relative to the resonance frequency of the protons of the reference substance. In other words, the frequency difference is measured directly. The standard reference substance that is used universally is tetramethylsilane, (CH3)4Si, also called TMS. This compound was chosen initially because the protons of its methyl groups are more shielded than those of most other known compounds. At that time, no compounds that had better-shielded hydrogens than TMS were known, and it was assumed that TMS would be a good reference substance since it would mark one end of the range. Thus, when another compound is measured, the resonances of its protons are reported in terms of how far (in Hertz) they are shifted from those of TMS.
The shift from TMS for a given proton depends on the strength of the applied magnetic field. In an applied field of 1.41 Tesla the resonance of a proton is approximately 60 MHz, whereas in an applied field of 2.35 Tesla (23,500 Gauss) the resonance appears at approximately 100MHz.The ratio of the resonance frequencies is the same as the ratio of the two field strengths:
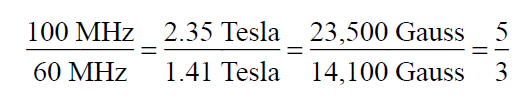
Hence, for a given proton, the shift (in Hertz) from TMS is 5/3 larger in the 100-MHz range (Bo=2.35 Tesla) than in the 60-MHz range (Bo=1.41 Tesla). This can be confusing for workers trying to compare data if they have spectrometers that differ in the strength of the applied magnetic field. The confusion is easily overcome if one defines a new parameter that is independent of field strength-for instance, by dividing the shift in Hertz of a given proton by the frequency in megahertz of the spectrometer with which the shift value was obtained. In this manner, a field-independent measure called the chemical shift
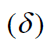
is obtained.

The chemical shift in
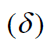
units express the amount by which a proton resonance is shifted from TMS, in parts per million (ppm), of the spectrometer’s basic operating frequency.