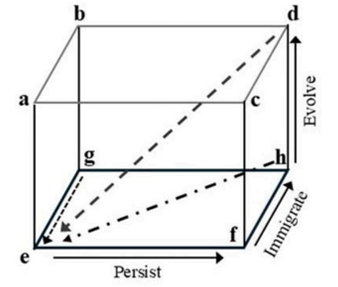
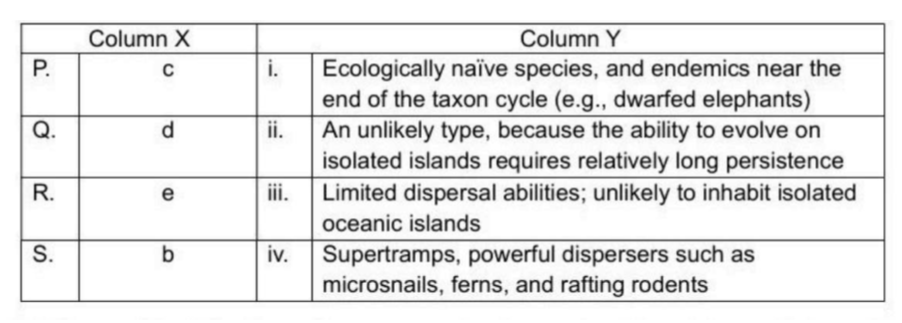
Correct option is C
The structure of a water (H₂O) molecule involves an oxygen atom covalently bonded to two hydrogen atoms, and it has the following characteristics:
Hybridization of oxygen: The oxygen atom in a water molecule undergoes sp³ hybridization. This means that the oxygen's 2s and three 2p orbitals mix to form four equivalent sp³ hybrid orbitals. These hybrid orbitals are used to form two sigma bonds with the hydrogen atoms and accommodate two lone pairs of electrons.
Bond angle (H-O-H): The theoretical bond angle for sp³ hybridization in a perfect tetrahedron is 109.5°. However, because of the presence of the lone pairs on the oxygen atom, the bond angle in the water molecule is slightly reduced. The actual H-O-H bond angle in water is 104.5°, which is slightly smaller due to the lone pairs of electrons on oxygen, which exert greater repulsive forces compared to bonding pairs.
Information Booster:
sp³ Hybridization: In the case of the water molecule, oxygen's sp³ hybridization occurs because it forms two single bonds with hydrogen atoms and has two lone pairs of electrons. These lone pairs take up more space, which slightly compresses the bond angle from the ideal 109.5°.
Bond Angle of 104.5°: The bond angle is reduced from the ideal tetrahedral angle due to the repulsion between the lone pairs of electrons. Lone pairs of electrons occupy more space than bonding pairs because they are localized on the nucleus, pushing the bonded hydrogen atoms closer together.
Additional Knowledge:
- sp² Hybridization (Incorrect Option 2 and 4):
- In sp² hybridization, there are three hybrid orbitals that form bonds with atoms, typically seen in molecules with double bonds (e.g., in alkenes or formaldehyde), and results in a 120° bond angle. This is not applicable to water since water does not have double bonds or sp² hybridization.
- Tetrahedral Geometry: The geometry of the water molecule is often described as bent or V-shaped due to the lone pairs on oxygen. Although the molecule has a tetrahedral arrangement of electron pairs around the oxygen atom, the bonding angle is altered due to the repulsion between lone pairs.