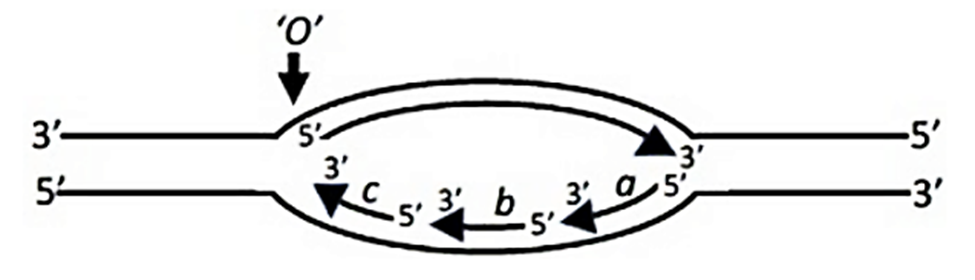
Correct option is C
Hoogsteen base pairing is an alternative base-pairing geometry different from the standard Watson-Crick model. It occurs when a purine (A or G) adopts a syn conformation instead of the usual anti conformation and pairs with a pyrimidine in the anti conformation.
Analyzing Each Option:
(anti A base-paired with anti T) - Incorrect
- In Watson-Crick base pairing, both adenine (A) and thymine (T) are in the anti conformation.
- Hoogsteen pairing requires syn-anti conformation.
(anti G base-paired with anti C) - Incorrect
- Watson-Crick G-C pairing occurs in the anti-anti conformation.
- Hoogsteen pairing is not observed in this form.
(syn A base-paired with anti T) - Correct
- This represents a classical Hoogsteen A-T pairing, where:
- Adenine (A) adopts the syn conformation.
- Thymine (T) remains in the anti conformation.
- This occurs under conditions such as low pH or protein binding and plays a role in DNA-protein interactions.
- This represents a classical Hoogsteen A-T pairing, where:
(anti G base-paired with anti U) - Incorrect
- Hoogsteen pairing in guanine requires a syn-anti configuration, not anti-anti.
- Guanine can form Hoogsteen bonds, but not in this conformation.
Information Booster:
Hoogsteen Base Pairing
- An alternative hydrogen bonding pattern between purines and pyrimidines.
- Found in triplex DNA, DNA-protein complexes, and damaged DNA sites.
Syn vs. Anti Conformations
- Anti: Standard orientation in Watson-Crick pairing.
- Syn: Flipped purine orientation leading to Hoogsteen pairing.
Examples of Hoogsteen Base Pairs
- A (syn) – T (anti)
- G (syn) – C+ (anti) (Protonated cytosine, seen in low pH).
Biological Importance
- Facilitates DNA flexibility and protein-DNA interactions.
- Helps in triplex DNA formation (important in gene regulation).
Conditions Favoring Hoogsteen Pairing
- Low pH (acidic environment).
- DNA bound to proteins.
- Certain drug-DNA interactions.