Correct option is D
Amphoteric oxides are oxides which have properties of both acidic and basic oxides. The property is known as amphoterism. Generally non-metallic oxides are acidic and metallic oxides are basic. Some non-metallic oxides are neutral and some metallic and semi-metallic oxides are amphoteric.
As these oxides can act as either an acid or a base, they can neutralize both acids and bases. For example, aluminium oxide (Al2O3) reacts with hydrochloric acid to form aluminium chloride and water. With sodium hydroxide solution it reacts into sodium aluminate (NaAlO2) and water.
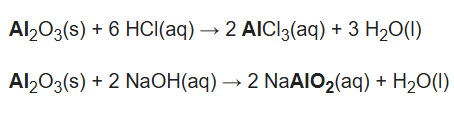
Tin(IV) oxide, also known as stannic oxide, is the inorganic compound with the formula SnO2. Although SnO2 is insoluble in water, it is amphoteric, dissolving in base and acid.
Tin oxides dissolve in acids. Halogen acids attack SnO2 to give hexahalostannates, such as [SnI6]2−. One report describes reacting a sample in refluxing HI for many hours.
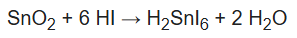
Similarly, SnO2 dissolves in sulfuric acid to give the sulfate:

The latter compound can add additional hydrogen sulfate ligands to give hexahydrogensulfatostannic acid.
SnO2 dissolves in strong bases to give "stannates," with the nominal formula Na 2 SnO 3 Dissolving the solidified SnO2/NaOH melt in water gives Na2[Sn(OH)6], "preparing salt," which is used in the dye industry.
Additional Knowledge
CaO (Calcium oxide): Calcium oxide (formula: Ca O), commonly known as quicklime or burnt lime, is a widely used chemical compound. It is a white, caustic, alkaline, crystalline solid at room temperature. It reacts with water to form calcium hydroxide and does not exhibit amphoteric properties.
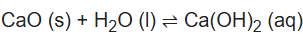





 English
English 150 Questions
150 Questions 150 Mins
150 Mins

