Correct option is A
The thermal stability of the alkali metal carbonate increases on moving down the group in the order:
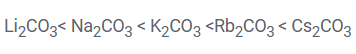
The thermal stability of these carbonates is influenced by the size and charge of the cation. Larger cations stabilize the carbonate ion more effectively because they produce a lower charge density, reducing the polarization effect on the carbonate ion. As a result, the carbonate ion is less likely to decompose into CO2 and the corresponding metal oxide.
On moving down the group, the size of the cation increases and the polarising power decreases. Hence, the stability of carbonate increases.
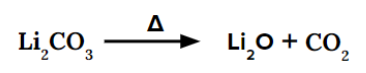
Exceptionally, on heating, lithium carbonate quite easily decomposes to give lithium oxide (Li2O) and carbon dioxide (CO2).
Li+ ion has a high charge density (charge/volume) due to its small size. So, it has a high polarising power and cannot stabilise the large polarizable CO32- ion. That is why it is unstable and readily decomposes on heating.





 English
English 150 Questions
150 Questions 150 Mins
150 Mins

