Correct option is B
Potassium superoxide is an inorganic compound with the formula K O2. It is a yellow paramagnetic solid that decomposes in moist air. It is a rare example of a stable salt of the superoxide anion. It is used as a CO 2 scrubber, H 2 O dehumidifier, and O 2 generator in rebreathers, spacecraft, submarines, and spacesuits.
Potassium superoxide is produced by burning molten potassium in an atmosphere of excess oxygen.
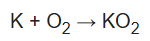
The salt consists of K+ and O2- ions, linked by ionic bonding.
Additional Knowledge
Potassium oxide (K 2 O) is an ionic compound of potassium and oxygen. It is a base. This pale yellow solid is the simplest oxide of potassium. It is a highly reactive compound that is rarely encountered. Some industrial materials, such as fertilizers and cements, are assayed assuming the percent composition that would be equivalent to K2O.
Potassium oxide is produced from the reaction of oxygen and potassium; this reaction affords potassium peroxide, K2O2. Treatment of the peroxide with potassium produces the oxide:
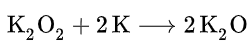
Potassium peroxide is an inorganic compound with the molecular formula K2O2. It is formed as potassium reacts with oxygen in the air, along with potassium oxide (K2O) and potassium superoxide (KO2).





 English
English 150 Questions
150 Questions 150 Mins
150 Mins

