Correct option is D
Copper extraction process: Chalcopyrite (CuFeS2), commonly known as copper pyrite or copper iron sulphide, is the most common ore utilised in copper extraction.
1) Concentration of ore: In this process, the ore is crushed to a fine powder. Then, the mineral is separated from the other particles using the froth flotation method. This method separates hydrophobic from hydrophilic materials to extract the mineral ore by increasing its concentration.
2) Roasting: In a reverberatory furnace, concentrated ore and SiO2 are heated in excess of air.
Volatile impurities are removed in this step. The sulphide is heated in the presence of air in a free supply of air below m.p. Sulphur, phosphorus, arsenic and antimony impurities are transformed to their corresponding volatile oxides and subsequently eliminated.
3)
Slag formation: Ore that has been roasted is mixed with sand before being burned in a furnace.
Slag available in the upper layer is removed. The underlying layer, which is mostly cuprous sulphide, Cu2S (98%), with a small quantity of iron sulphide, FeS (2%), is referred to as matte.
4) Bessemerisation: This is the process of blowing air into a molten copper mat that has been placed in a Bessemer converter. The remaining products, including FeS, are oxidised and removed as slag in the last step of smelting (FeSiO3).
The Impure copper that has a blister-like appearance is referred to as blister copper.
5) Poling: To convert any copper oxide contaminant into Cu, molten Cu is agitated with green wood poles.
Lead extraction process: Metallic lead can be extracted from lead sulfide (PbS) by oxidation–reduction processes or by direct oxidation to metal, i.e., roast reaction. Oxidation–reduction processes include the two stages, sinter plant–blast furnace process and the direct smelting-reduction processes.
Sinter Plant-Blast Furnace
In this path, lead sulfides are oxidized in the solid state to remove sulfur while producing PbO. The sinter product (PbO) is then reduced to lead, typically in a blast furnace charged with coke. Flux is also added to form a molten slag in which the iron oxides are removed.
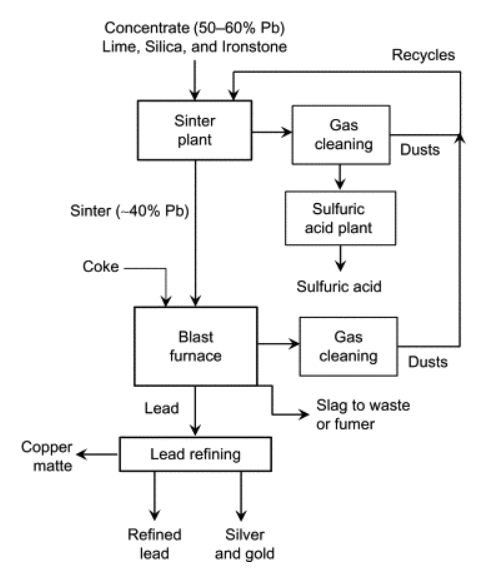
The main problems with the sintering-blast furnace route are emissions of particulate lead and greenhouse gases, little use of the exothermic heat of the oxidation during sintering, and use of metallurgical coke, the production of which contributes to the emissions of greenhouse gases, in particular, carbon dioxide.
Direct Smelting Reduction
This route takes place at sufficient high temperatures (>1473 K) to melt the oxidation products. Both oxidation and reduction steps take place in a single unit. This allows the heat of the sulfide oxidation to be utilized directly in the process.
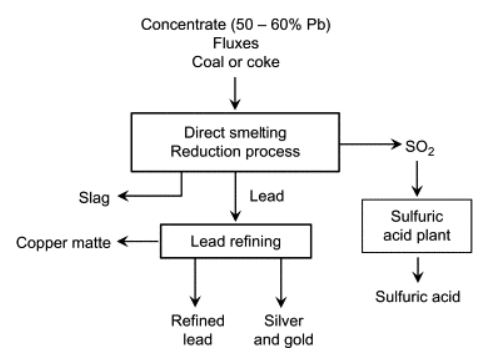
Roast Reaction
This route is based on the roast reactions and can be considered as one of the first smelting paths used for lead production. In this route, lead sulfide is partially oxidized by air to produce metallic lead, PbO, and PbSO4, which collect at the furnace bottom, forming a molten slag layer at the top of liquid lead. In the molten bath, the desulfurization process continues by the roast reactions, i.e., lead oxide and sulfate react with the remaining lead sulfide yielding metallic lead. This path requires high-grade lead concentrates and produces a small amount of slag that contains a high level of lead oxide to be subsequently reduced by carbon.
Additional Knowledge
Iron extraction process: Iron is extracted from iron ore using carbon and limestone inside a blast furnace. Iron ores are present as iron oxides in rocks like magnetite and haematite. The iron is usually pelletised before being transported to a blast furnace, where the iron is extracted using a reduction reaction where limestone and coke (made from coal) react with heat to remove oxygen from the iron oxide in the iron ore.





 English
English 150 Questions
150 Questions 150 Mins
150 Mins

