Correct option is D
The correct answer is (d) and .
The two naturally occurring isotopes of chlorine are:
Chlorine-35 (_17^35 Cl): This isotope contains 17 protons and 18 neutrons.
Chlorine-37 (_17^37 Cl): This isotope contains 17 protons and 20 neutrons.
These isotopes have the same number of protons (17), which makes them chlorine, but they differ in the number of neutrons, leading to different atomic masses. Chlorine-35 accounts for about 75% of natural chlorine, while Chlorine-37 makes up the remaining 25%.
Information Booster:
Isotopes: Are variants of the same element that have the same number of protons but different numbers of neutrons.
Chlorine-35 and Chlorine-37: These two isotopes combine to give chlorine an average atomic mass of approximately 35.5.





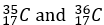
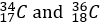
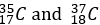
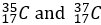
 English
English 10 Questions
10 Questions 10 Mins
10 Mins

