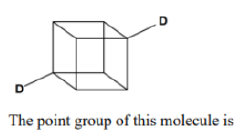
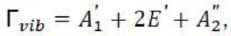Correct option is C
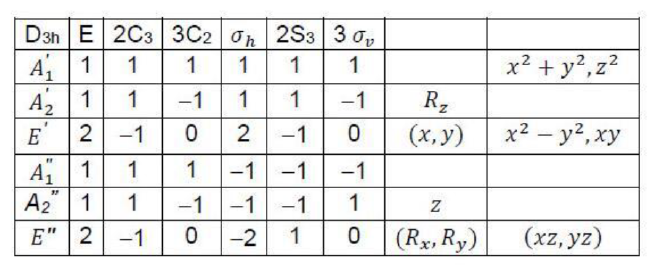
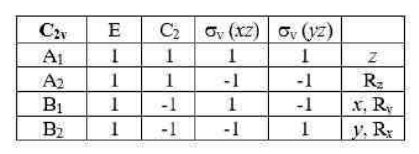
In molecular symmetry, the character table provides a way to represent the symmetries of the vibrational modes, electronic states, and other molecular properties. For the C3v point group (as seen in ammonia, NH3 ), the irreducible representations (A1, A2, and E) describe how a particular mode transforms under the symmetry operations of the group.
• Irreducible Representations: The symmetries of the different normal modes are classified into these irreducible representations, which can combine to describe higher energy states.
• First Excited State: The symmetry of the first excited state of NH3 is E, a doubly degenerate representation. The transition to higher excited states involves combining multiple irreducible representations.
In quantum systems, the second excited state can be derived by analyzing the direct product of the irreducible representations of the system. For the E mode:
E⊗E=A1 +A2 + E
This result indicates that when the mode is excited a second time, its symmetry components split into A1 , A2 and E irreducible representations.