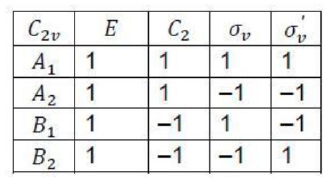
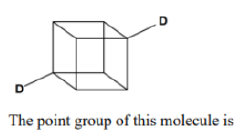
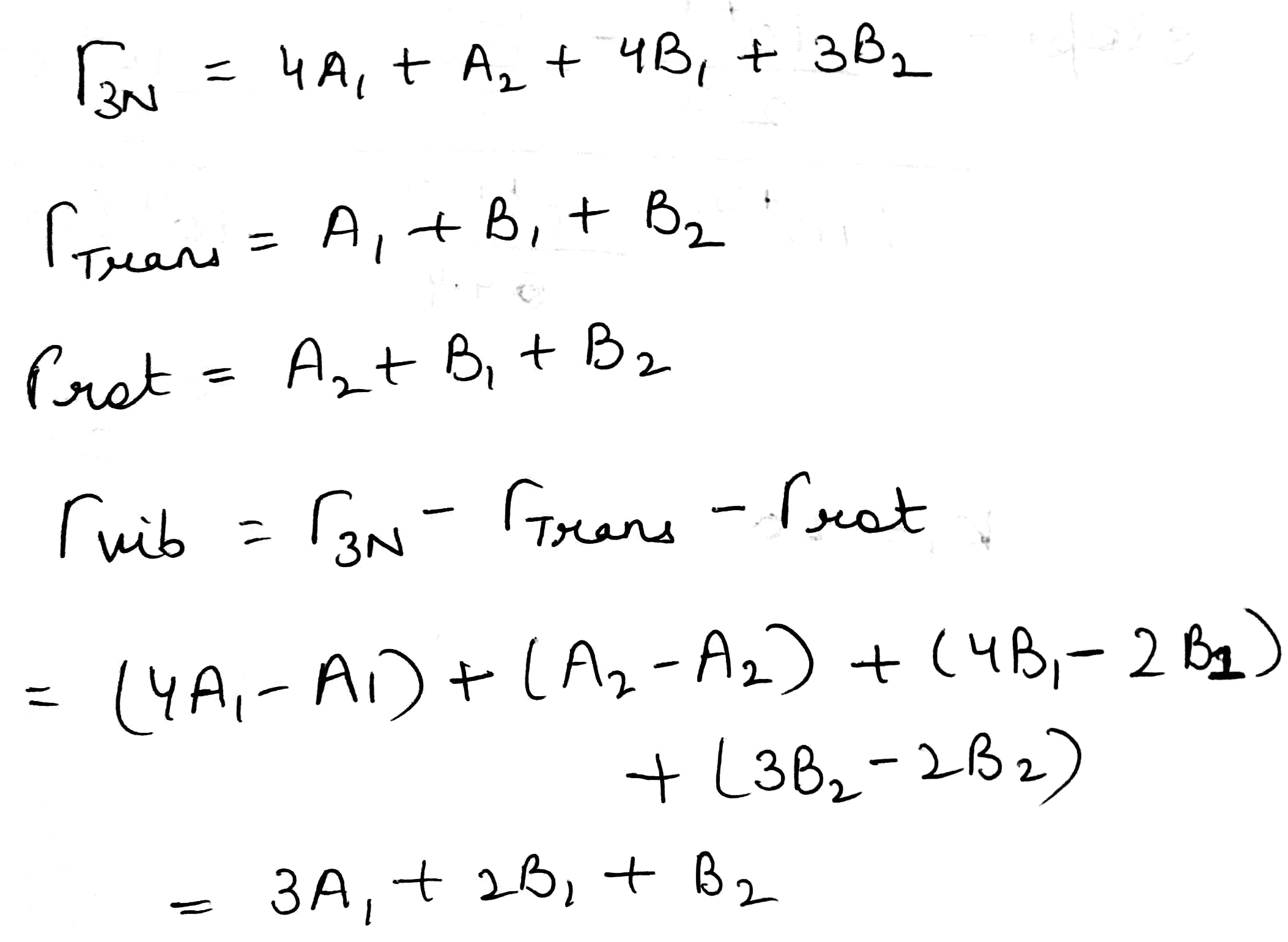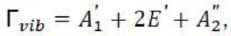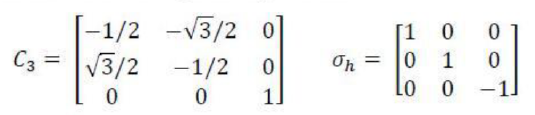Correct option is B
In a normal mode vibration, each atom is displaced from its equilibrium position by a vector that can but need not lie along the bond direction (for example in a bending mode). The directions and magnitudes of the displacements are not the same for all atoms.
How many normal modes does a molecule have? An isolated atom has three translational degrees of freedom; therefore, a molecule consisting of n atoms has 3n degrees of freedom. Three of these are translations of the molecule and are not of interest here. A nonlinear molecule with n atoms has three degrees of rotational freedom, and the remaining 3n-6 internal degrees of freedom correspond to normal modes of vibration. Because a linear molecule has only two degrees of rotational freedom, it has 3n-5 normal modes of vibration.
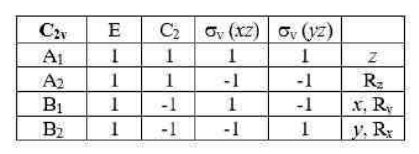
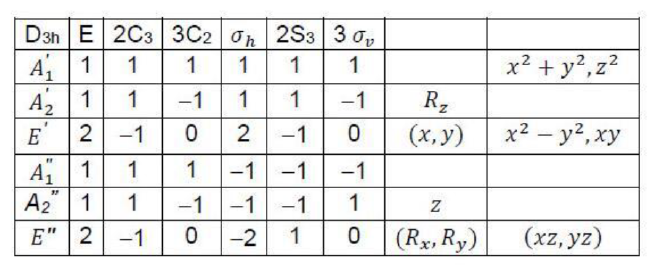
The translation of the molecules along the x, y, and z axes as well as their rotation about the same axes must be separated out to obtain the representations of the vibrational normal modes. This can be done by subtracting the representations belonging to x, y, and z as well as to Rx, Ry, and Rz.