Correct option is A
In crystallography, the separation between planes in a crystal lattice is determined using Bragg's law and the Miller indices (h, k, l) of the planes. For a face-centered cubic (FCC) crystal, the planes with the lowest Miller indices typically have the smallest spacing, and the spacing between planes is inversely proportional to the square root of the sum of the squares of the Miller indices.
Interplanar Spacing Formula: The separation (dhkl) between planes in a cubic crystal is given by the formula:
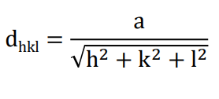
where (a) is the unit cell length and (h, k, l) are the Miller indices of the planes.









