Correct option is A
Explanation-
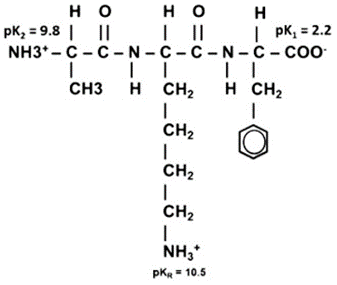
Step 1: Identify the ionizable groups and their pKa values
From the image, the peptide has three ionizable groups:
α-carboxyl group (C-terminal): pKa₁ = 2.2
α-amino group (N-terminal): pKa₂ = 9.8
Side chain of lysine (–NH₃⁺): pKaR = 10.5
Step 2: Determine the charge states at different pH
At low pH (e.g., pH 1):
All ionizable groups are protonated:
NH₃⁺ (N-term): +1
COOH (C-term): 0
Lysine side chain NH₃⁺: +1
→ Net charge = +2
As pH increases:
First, the C-terminal COOH loses a proton at pKa ≈ 2.2 → becomes COO⁻ (charge −1)
→ Net charge = +1
Next, the N-terminal NH₃⁺ loses a proton at pKa ≈ 9.8 → becomes NH₂ (charge 0)
→ Net charge = 0 . This is one potential point of isoelectricity
Then, the lysine side chain NH₃⁺ loses a proton at pKa ≈ 10.5 → becomes NH₂ (neutral)
→ Net charge = −1
Step 3: Find the pI (pH at which net charge = 0)
The pI lies between the two pKa values where the peptide has a net charge of +1 and −1 — that is, between:
pKa₂ = 9.8 (N-terminal NH₃⁺ → NH₂)
pKaR = 10.5 (Lysine side chain NH₃⁺ → NH₂)
So,
Correct answer: Option a : 10.15