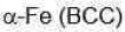Correct option is B
A crystal is built up from regularly repeating ‘structural motifs’, which may be atoms, molecules, or groups of atoms, molecules, or ions. A space lattice is the pattern formed by points representing the locations of these motifs. The unit cell is an imaginary parallelepiped (parallel-sided figure) that contains one unit of the translationally repeating pattern. A unit cell is commonly formed by joining neighbouring lattice points (A lattice in the sense of a 3-dimensional array of regularly spaced points coinciding with e.g. the atom or molecule positions in a crystal) by straight lines. Such unit cells are called primitive. A primitive unit cell (with lattice points only at the corners) is denoted P. A face-centred unit cell (F) has lattice points at its corners and also at the centres of its six faces.
Designation of planes
In order to discuss the structure of a crystal, we need to describe the orientation of planes passing through lattice points of the crystal. The orientation of a lattice plane can be described by considering the intercepts of the plane on the three basis vectors of the lattice. According to Haüy, it is possible to choose the unit lengths a, b and c along the three basis vectors such that the ratio of each of the intercepts h', k' and l' to the corresponding unit length is either an integer or a ratio of two integers. This statement is known as the law of rational indices. The three ratios are known as the Weiss indices of the plane.
Instead of using Weiss indices, it is more advantageous to use the Miller indices. In the latter, we take the reciprocal of the three Weiss indices and then multiply them by the smallest number (if necessary) to make them all integers. The three resultant integers are known as Miller indices and are represented as (hkl).
The distances between planes of a crystal can be determined with the help of X-ray diffraction measurements. These can, in turn, be used to determine the system to which the crystal belongs.
When X-rays are incident on a crystal face, they penetrate into the crystal and suffer reflections on striking the atoms in successive planes. If the reflected waves from successive layers are out of phase, then due to the destructive interference, no diffraction will be observed. If, however, the reflected waves are in phase, then due to the constructive interference, a diffraction spot will be observed. The condition for a reflection to give constructive interference.
Let θ be the incident angle of monochromatic X-rays of wavelength λ with the parallel equidistant planes of atoms with interplanar spacing equal to d. The waves are in phase before striking the planes. Two such waves labelled as wave 1 and wave 2. After the reflection, the two waves will be in phase provided the extra distance travelled by wave 2 is an integral multiple of wavelength λ. This extra distance can be obtained by dropping perpendiculars BG and BH from B on to wave 2.
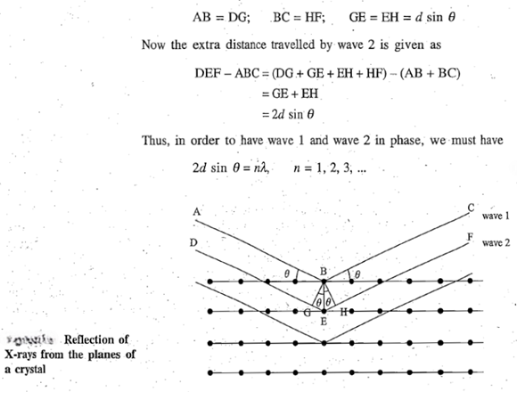
The constant n gives the order of reflection and is equal to the number of wavelengths in the path difference between waves reflected by adjacent planes. Thus, n can take only integral values subject to the limitation that sin θ cannot be greater than one. For fixed values of λ and d, we can have more than one reflection at angles θ1, θ2... corresponding to the values of n equal to 1, 2, 3,..., respectively. It can be concluded that the higher order reflections will occur at larger values of sin θ and hence at larger angles. Experimentally, it is found that the lower order reflections are the most intense and the intensities of higher order reflections decreases rapidly.
For a body-centred cubic lattice, all diffraction lines for which (h+k+l) is an odd integer must be absent.