Correct option is D
To interpret the polypeptide fold from the circular dichroism (CD) spectra, we need to examine both:
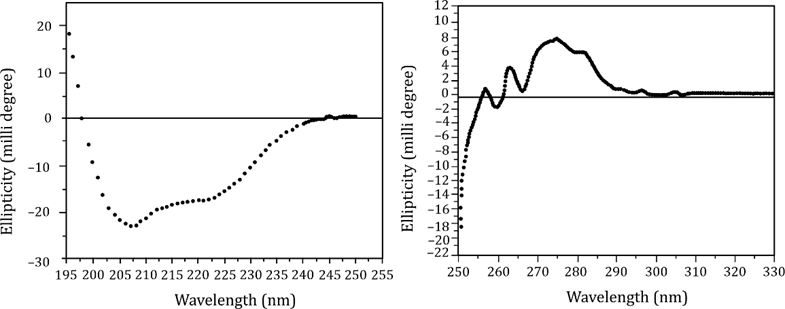
Far-UV CD spectrum (left graph): Provides information on secondary structure.
Near-UV CD spectrum (right graph): Provides information on tertiary structure and environment of aromatic residues.
Far-UV CD (195–250 nm):
Shows a broad negative band near 218 nm, and a positive band near 195 nm.
This pattern is typical of β-sheet structure.
α-helix would show double minima at ~208 nm and ~222 nm — not observed here.
So the secondary structure is predominantly β-sheet.
Near-UV CD (250–320 nm):
Shows distinct signals, especially between 270–290 nm, indicating:
Well-packed tertiary structure
Aromatic side chains (like Tyr, Trp, Phe) are in an ordered, asymmetric environment
This suggests a stable 3D fold, often seen in proteins with both α and β domains, or complex tertiary folds.
Interpretation of options:
It contains only β-sheets
Too strict. Near-UV data implies more complex folding.It has to be an alternate α/β fold
No α-helix evidence in far-UV. This needs alternating α-helix and β-sheet — not supported here.It has to be a mixed α+β fold
No α-helix in the far-UV spectrum — again, ruled out.It belongs to either alternate α/β or mixed α+β fold
This is the most inclusive and correct answer, considering:Far-UV: Strong β-sheet signal
Near-UV: Structured tertiary fold, suggesting potential for both α and β regions
Could be a mixed fold or α/β arrangement that’s not fully evident in far-UV due to weak α content