Correct option is C
EXPLANATION-
Step 1: Identify a data point from the graph
Looking at the graph:
When concentration
Absorbance
Step 2: Use Beer-Lambert Law to find
So, the correct option is (c) 33.3
The absorbance values of a dye measured at 600 nm were plotted against its corresponding concentrations, as given below.
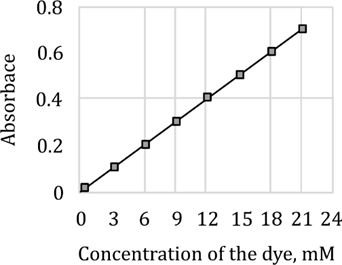
Which of the following will be the best estimate of the extinction coefficient of the compound in units? The path length of the cuvette used for the measurement is 1 cm.
EXPLANATION-
Step 1: Identify a data point from the graph
Looking at the graph:
When concentration
Absorbance
Step 2: Use Beer-Lambert Law to find
So, the correct option is (c) 33.3
Which one of the following techniques CANNOT be used for separation, detection, or visualization of DNA?
Which one of the following is the most sensitive immunoassay?
Optical remote sensing has been increasingly used to monitor vegetation globally. The table below lists different regions of the electromagnetic radiation (EMR) spectrum as well as different vegetation characteristics:
List I | List II |
Part of the EMR spectrum | Vegetation characteristics |
A. Ultraviolet | I. Plant water content |
B. Visible | II. Foliage density |
C. Near Infrared | III. Plant photosynthetic pigments |
D. Shortwave Infrared |
Which one of the following combinations correctly matches the EMR region with the vegetation character analysed:
To delineate the steps in endoplasmic reticulum (ER) transport, a PhD student homogenized pancreatic acinar cells to isolate microsomes, which retain most of the biochemical properties of the ER. For this experiment, the student has planned a number of controls as mentioned below.
A. Treat one set of microsomes first with detergent and then with protease.
B. Treat one set of microsomes with protease only.
C. Treat one set of microsomes with micrococcal nuclease.
D. Treat one set of microsomes with detergent only.
Select the option that represents the best combination of the controls.
The pI of four proteins (A, B, C, D) are shown in the table below:
Protein | pI |
A | 4 |
B | 5.5 |
C | 7 |
D | 4 |
To purify 'D' from a mixture of these four proteins in a single step, using ion-exchange chromatography, what combination of buffer pH and ion-exchange resin would be used?
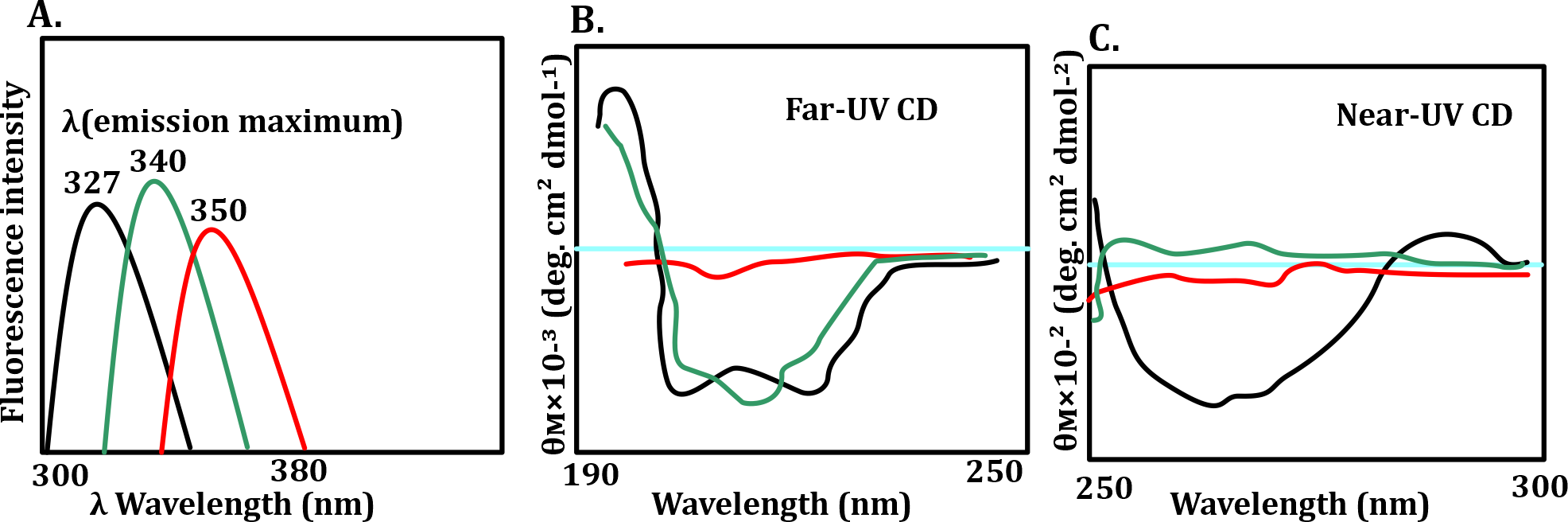
A circular dichroism spectrum in the far-UV region informs on the kind and content of secondary structures in a protein. Near-UV and tryptophan emission spectra inform on the tertiary structure. Shown in the panels above are (A) Intrinsic fluorescence emission spectra of protein 'X', (B) Far-UV CD spectra of protein 'X', (C) Near-UV CD spectra of protein 'X' recorded under different conditions.
Curves represent the spectra of protein 'X' at pH 7.0 (black), pH 3.0 (green), and pH 7.0 in the presence of 6.0 M guanidine hydrochloride (red).
What does the experiment report?
What will be the percentage transmission when absorbance is 1, 2, and 3 respectively?
The diffraction patterns of two forms of DNA (A and B) and statements related to these patterns are given below.

A. The diffraction pattern corresponding to A-DNA is shown by form 1 with 11.6 base pairs per turn.
B. The diffraction pattern of B-DNA is shown by form 2 with 11.6 base pairs per turn.
C. The diffraction pattern of A-DNA is shown by form 2 with 11.6 base pairs per turn.
D. The diffraction pattern of B-DNA is shown by form 1 with 10 base pairs per turn.
Which one of the following options have all correct statements?
Which one of the following correctly describes the spectroscopic experiment that would help distinguish between a α helix, a 3₁₀ helix and a π helix?
In a conjugation experiment between bacterial Hfr strain ‘X’ and F⁻ cell, lac gene enters the recipient in 4 minutes, but the F⁻ cells remain auxotrophic for Leu, Trp, Ura, Glu, Phe, and Gly. The mating is then allowed to proceed for 20 minutes and lac⁺ exconjugants are selected. Of the lac⁺ cells:
35% are leu⁺
98% are trp⁺
10% are ura⁺
65% are glu⁺
0% are phe⁺
81% are gly⁺
Select the correct order of the genes as they enter, from the choices given below:

