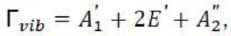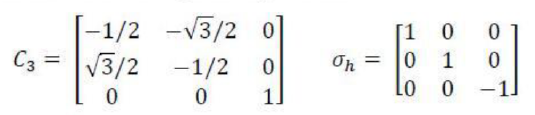Correct option is C
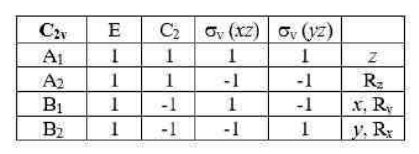
The symmetry number or symmetry order of an object is the number of different but indistinguishable (or equivalent) arrangements (or views) of the object, that is, it is the order of its symmetry group. The object can be a molecule, crystal lattice, lattice, tiling, or in general any kind of mathematical object that admits symmetries.
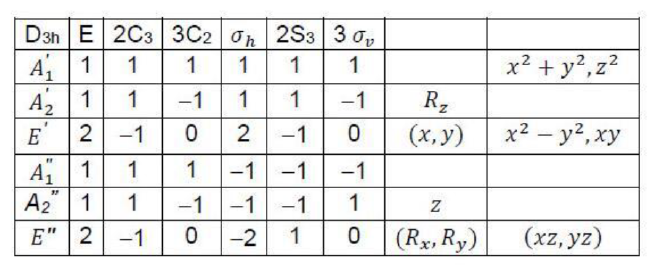
The symmetry operations of BCl3 molecule are:
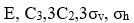
and
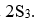
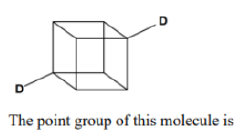
. The point group of
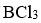
is
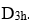
. The number of equivalent structures is given by n-1, where, n is the order of the point group. The order is always an integer multiple of n of the principal axis.
Rotational subgroup in the question corresponds to axis of rotation. The symmetry number of BCl3 can be calculated as:
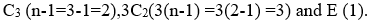
Thus, the answer is 2+3+1=6.