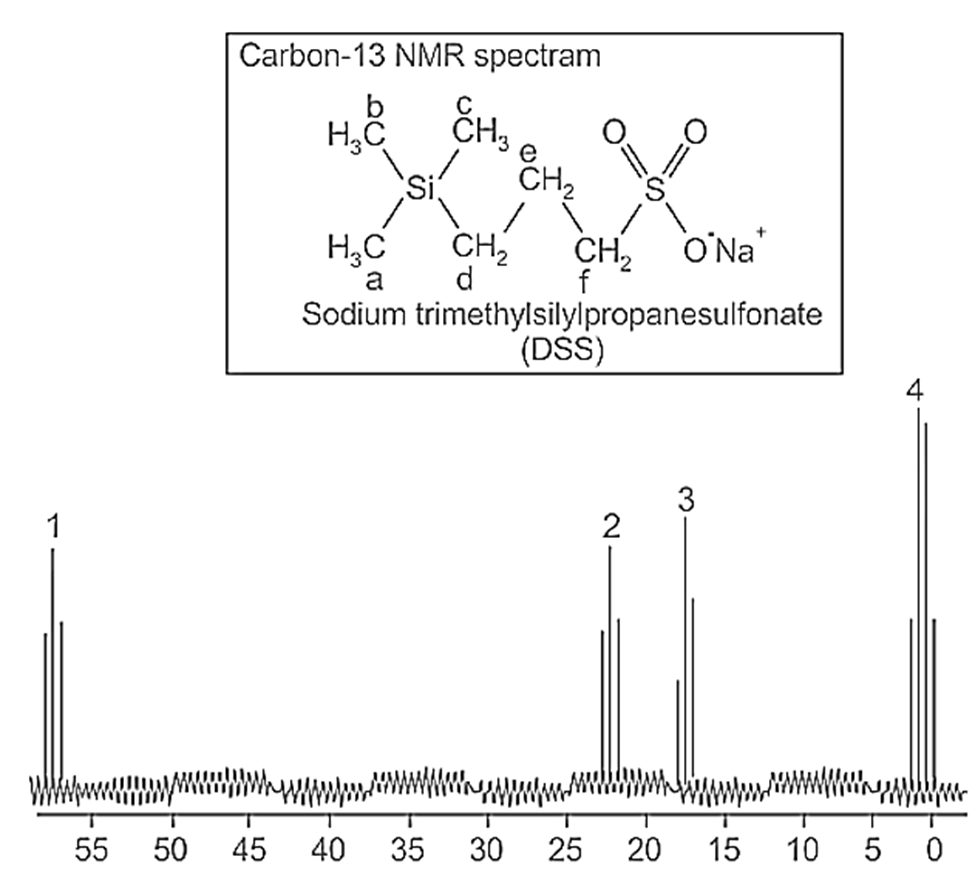
Correct option is B
Upon reviewing the spectrum and DSS structure:
Carbon a corresponds to the silicon-bound carbon (part of the trimethylsilyl group), which typically shows a peak at around 0-10 ppm.
Carbon b is the methyl carbon (-CH₃) attached to silicon, usually observed near 0-10 ppm.
Carbon c is the carbon in the propyl chain connected to the sulfonate group, which is generally observed between 20-30 ppm.
Carbons d, e, and f are part of the remaining carbons in the propyl chain attached to the sulfonate group and usually appear in the region between 30-50 ppm.
The assignment of peaks in Option 2 is correct:
Peak 1 corresponds to carbon f, which is part of the propyl chain. In the spectrum, this peak is relatively upfield and corresponds to the carbon that is farther away from the electronegative sulfonate group.
Peak 2 corresponds to carbon e, which is also part of the propyl chain but slightly more downfield than carbon f.
Peak 3 corresponds to carbon d, located near the sulfonate group in the propyl chain.
Peak 4 corresponds to carbons a, b, and c, which are associated with the silicon-bound group and the propyl chain's terminal group (attached to the sulfonate).
This matches the expected patterns for DSS and the chemical shifts of each carbon group in the spectrum.
Additional Information
Option 1: Incorrect. This option incorrectly places carbon a at peak 1, which corresponds to a more shielded region of the spectrum and should not be associated with peak 1, which is instead assigned to carbon f.
Option 3: Incorrect. The assignment of carbon d to peak 1 and carbon f to peak 3 is inconsistent with their relative positions in the structure of DSS and their expected chemical shifts.
Option 4: Incorrect. The assignment of carbon e to peak 1 is incorrect because carbon e is part of the propyl chain, and it is more downfield than carbon f, which is placed in peak 1 in Option 2.