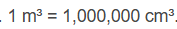Correct option is D
Crystal structure is a depiction of the orderly organization of atoms, ions, or molecules in a crystalline substance used in crystallography. The symmetry patterns that recur in the major directions of three-dimensional space in matter are caused by the inherent nature of the component particles, resulting in ordered structures.
The smallest collection of particles in the material that makes up this repeating pattern is the structure’s unit cell. The unit cell reflects the complete crystal’s symmetry and structure, which is built up via repeated translation of the unit cell along its fundamental axes.
The volume of the unit cell with monoclinic lattice can be calculated as:
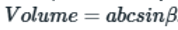
a, b and c are the lattice constants and beta is the angle greater than 90 degrees.
A unit cell’s density is defined as the ratio of its mass to its volume. The mass of the unit cell can be calculated by multiplying the total number of atoms present in the unit cell and the mass of each atom.
The mass of the unit cell can be written as: Mass= Z×m
Here, Z is the atomic number and m is the mass of the atoms.
The mass of the atoms present in the unit cell will be calculated as: