Correct option is C
The correct answer is Option (3):
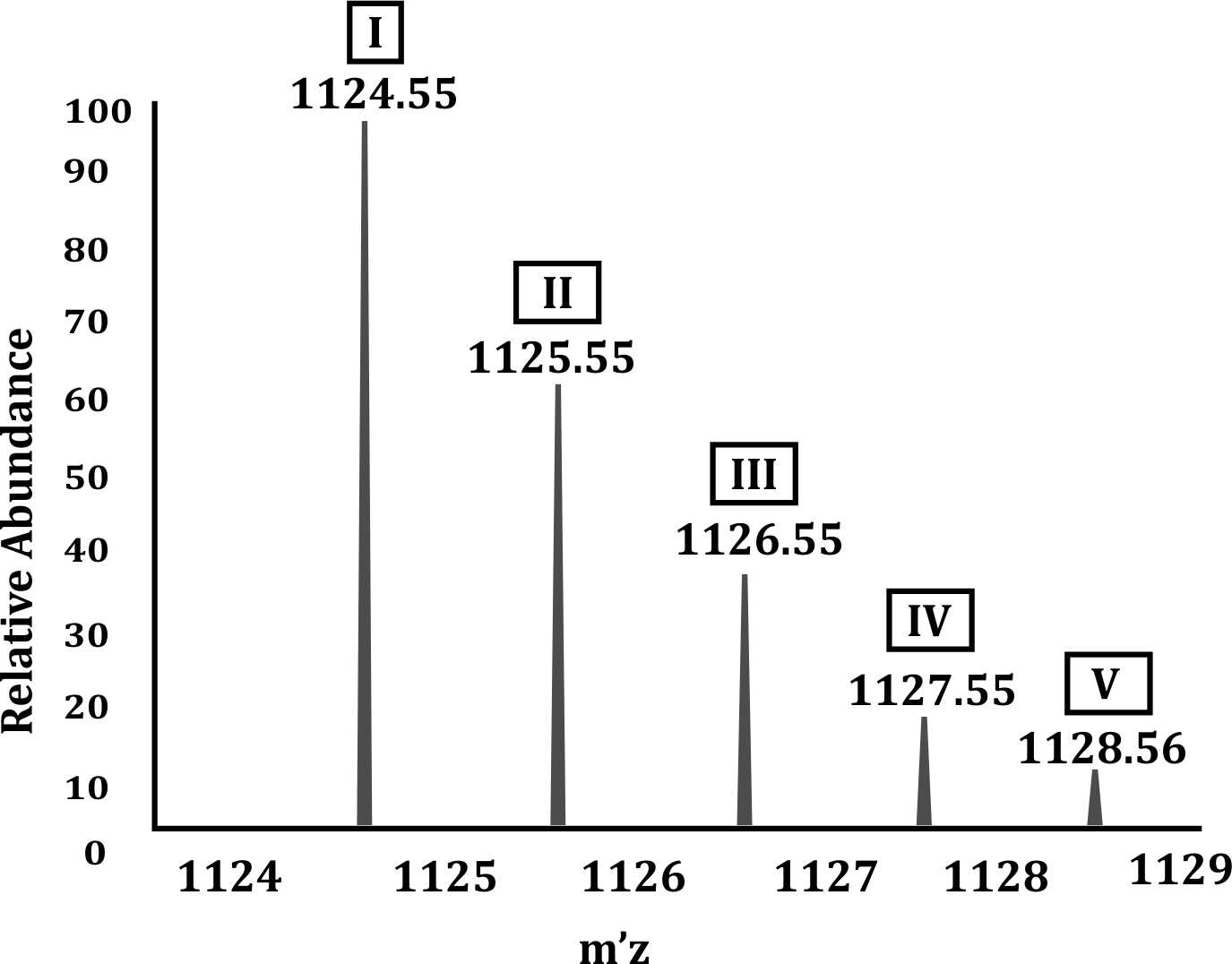
(A) ¹³C isotope distribution, (B) Peak I, and (C) 1124.55 Da
Explanation:
(A) Reason for multiple peaks in mass spectrum of a pure peptide:
- In mass spectrometry, a pure peptide typically gives multiple peaks due to the presence of isotopic variants, primarily due to ¹³C isotope distribution.
- Most atoms in nature are a mixture of isotopes, and for carbon, the natural abundance is about 98.9% ¹²C and 1.1% ¹³C.
- As a peptide contains many carbon atoms, the chance of incorporation of one or more ¹³C isotopes increases, leading to M+1, M+2, M+3, etc. peaks, which are observed as successive higher m/z peaks.
(B) Monoisotopic species:
- The monoisotopic peak (Peak I) represents the peptide with only the most abundant (lightest) isotopes, i.e., all atoms as ¹²C, ¹H, ¹⁴N, ¹⁶O, etc.
- Hence, the first peak (1124.55 m/z) is the monoisotopic peak, and it is typically the leftmost and often highest or one of the highest intensity peaks.
(C) Monoisotopic mass:
- The m/z value of Peak I = 1124.55 Da corresponds to the monoisotopic mass of the peptide, as it is calculated solely from the lowest mass isotopes.
Information Booster:
- Monoisotopic mass is the sum of the masses of the most abundant isotopes of all atoms in the molecule.
- Isotopic distribution (like ¹³C, ¹⁵N, etc.) causes a predictable series of M+1, M+2… peaks in the mass spectrum.
- The presence of ¹³C atoms increases the mass by 1 Da per ¹³C atom substituted.
- The relative intensity of these peaks depends on the number of carbon atoms and their probabilities of being replaced by ¹³C.
- In peptides with many carbon atoms, M+1 and M+2 peaks may sometimes be higher in abundance than M (monoisotopic).
- Mass spectrometry in positive ion mode typically detects [M+H]⁺ ions.
- The isotopic envelope helps in confirming molecular composition and verifying peptide purity.
Additional Information on Incorrect Options:
- Option 1 (Peak V as monoisotopic): Incorrect, Peak V is the M+4 peak, caused by multiple ¹³C substitutions.
- Option 2 and 4 (¹⁴C isotope distribution): ¹⁴C is a radioactive isotope, not naturally abundant, and does not cause this isotopic envelope in mass spectra.
- Hence, ¹³C is responsible, not ¹⁴C.