Correct option is A
Understanding the Potential Temperature Equation:
· Potential temperature (θ) is a thermodynamic parameter that represents the temperature an air parcel would have if it were brought adiabatically to a reference pressure P0.
· It is calculated using the formula
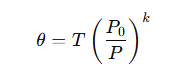
where:
· T is the actual temperature,
· P is the actual pressure,
· P0 is the reference pressure (often 1000 hPa),
· k=R/Cp.
k Definition:
· k=R/Cp:
· R is the specific gas constant for air.
· Cp is the specific heat at constant pressure.
· This ratio determines how the temperature changes with pressure during adiabatic processes.
· Explanation of Options:
· Option 1: Correct.
· k is defined as the ratio of the gas constant (R) to the specific heat at constant pressure (Cp) for the gas, which is ideal for air or diatomic gases.
· Option 2: Incorrect.
· k is not related to latent heat. Latent heat governs phase changes, not adiabatic processes.
· Option 3: Incorrect.
· Boltzmann constant (kB) is not part of this equation.
· Option 4: Incorrect.
· k is not related to height but rather thermodynamic properties.
Thus, the correct answer is Option 1: Ratio of gas constant to specific heat at constant pressure for ideal diatomic gas.
Information Booster: 1. Specific Heat and Gas Constant:
· Cp=Cv+R, where Cv is the specific heat at constant volume.
· For a diatomic gas like air:
· Cp=7/2 R, Cv=5/2R.
2. Adiabatic Processes:
· During adiabatic compression/expansion, the relationship between pressure and temperature is governed by k=R/Cp.
3. Applications of Potential Temperature:
· Useful in atmospheric science for analyzing vertical stability of the atmosphere.
· Helps identify layers with different thermodynamic properties





 English
English 10 Questions
10 Questions 12 Mins
12 Mins
 Choose the correct answer:
Choose the correct answer:


