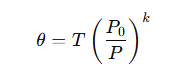Correct option is B
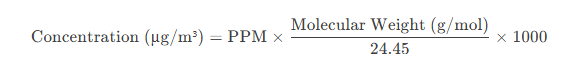
PPM= Parts per million (given as equal for all gases).
Molecular Weight= Molar mass of the gas (g/mol).
24.45= Molar volume (L/mol) at standard conditions (25°C and 1 atm).
1000= Conversion factor from grams to micrograms (µg).
Since PPM, temperature, and pressure are the same for all gases, the only variable affecting the µg/m³ concentration is the molecular weight of each gas.
The molecular weights of the given gases are:
CO₂ (Carbon Dioxide)= 44 g/mol
SO₂ (Sulfur Dioxide)= 64 g/mol
O₃ (Ozone)= 48 g/mol
NO₂ (Nitrogen Dioxide)= 46 g/mol
Since SO₂ has the highest molecular weight (64 g/mol), it will yield the highest µg/m³ concentration when converted from PPM.
SO₂ (Sulfur Dioxide) will have the highest concentration in µg/m³ when all gases are present at equal PPM levels.
Information booster
PPM (Parts Per Million): A volume-based unit representing the ratio of gas molecules to total air molecules.
Example: 1 PPM CO₂ = 1 CO₂ molecule per 1 million air molecules.Independent of molecular weight.
Used for regulatory standards (e.g., air quality limits).
µg/m³ (Micrograms per Cubic Meter): Amass-based unit representing the actual mass of a gas per unit volume of air.
Depends on themolecular weightof the gas.
Critical for health/toxicity assessments (e.g., SO₂ is harmful even at low µg/m³).





 English
English 10 Questions
10 Questions 12 Mins
12 Mins
 Choose the correct answer:
Choose the correct answer: