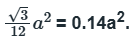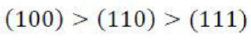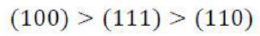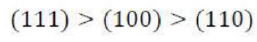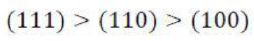Correct option is C
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals.
There are three main varieties of these crystals:
Primitive cubic (alternatively called simple cubic)
Body-centered cubic
Face-centered cubic
Note: the term fcc is often used in synonym for the cubic close-packed or ccp structure occurring in metals.
The face-centered cubic lattice has lattice points on the faces of the cube, that each gives exactly one half contribution, in addition to the corner lattice points, giving a total of four lattice points per unit cell (1⁄8 × 8 from the corners plus 1⁄2 × 6 from the faces).
Case 1:
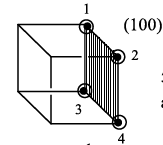
Five lattice points are present and the area is a2. The area occupied by one lattice point is
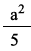
=0.20a2
Case 2:
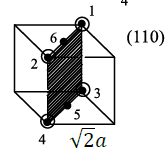
Six lattice points are present and the area is
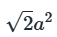
The area occupied by one lattice point is
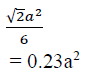
Case 3:
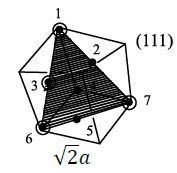
Six lattice points are present and the area is
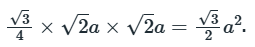
The area occupied by one lattice point is