Correct option is B
The valence band and conduction band are the bands closest to the Fermi level, and thus determine the electrical conductivity of the solid. In band structure theory, used in solid state physics to analyze the energy levels in a solid, the Fermi level can be considered to be a hypothetical energy level of an electron, such that at thermodynamic equilibrium this energy level would have a 50% probability of being occupied at any given time.
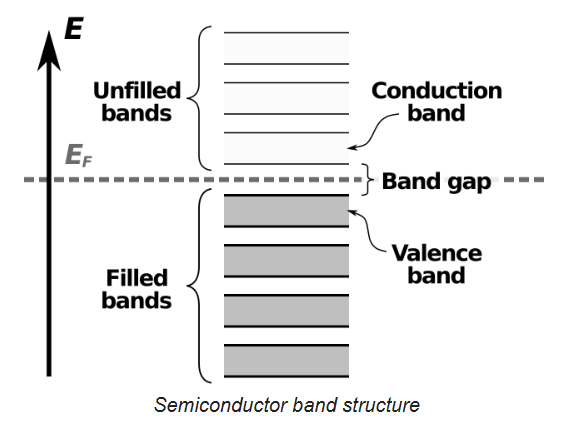
In nonmetals, the valence band is the highest range of electron energies in which electrons are normally present at absolute zero temperature, while the conduction band is the lowest range of vacant electronic states. On a graph of the electronic band structure of a semiconducting material, the valence band is located below the Fermi level, while the conduction band is located above it.
In semiconductors and insulators the two bands are separated by a band gap, while in conductors the bands overlap. A band gap is an energy range in a solid where no electron states can exist due to the quantization of energy. Within the concept of bands, the energy gap between the valence band and the conduction band is the band gap. Electrical conductivity of non-metals is determined by the susceptibility of electrons to be excited from the valence band to the conduction band.
Additional Knowledge
The Rydberg states of an atom or molecule are electronically excited states with energies that follow the Rydberg formula as they converge on an ionic state with an ionization energy. The Rydberg formula calculates the wavelengths of a spectral line in many chemical elements. Although the Rydberg formula was developed to describe atomic energy levels, it has been used to describe many other systems that have electronic structure roughly similar to atomic hydrogen. In general, at sufficiently high principal quantum numbers, an excited electron-ionic core system will have the general character of a hydrogenic system and the energy levels will follow the Rydberg formula. Rydberg states have energies converging on the energy of the ion. The ionization energy threshold is the energy required to completely liberate an electron from the ionic core of an atom or molecule.
An electron and an electron hole that are attracted to each other by electromagnetism can form a bound state called an exciton. It is an electrically neutral quasiparticle that exists mainly in condensed matter, including insulators, semiconductors, some metals, but also in certain atoms, molecules and liquids. The exciton is regarded as an elementary excitation that can transport energy without transporting net electric charge.
An exciton can form when an electron from the valence band of a crystal is promoted in energy to the conduction band e.g., when a material absorbs a photon. Promoting the electron to the conduction band leaves a positively charged hole in the valence band. Here 'hole' represents the unoccupied quantum mechanical electron state with a positive charge, an analogue in crystal of a positron. Because of the attractive coulomb force between the electron and the hole, a bound state is formed, akin to that of the electron and proton in a hydrogen atom or the electron and positron in positronium.
The small radius excitons, or Frenkel excitons, where the electron-hole relative distance is restricted to one or only a few nearest neighbour unit cells. Frenkel excitons typically occur in insulators and organic semiconductors with relatively narrow allowed energy bands and accordingly, rather heavy Effective mass.