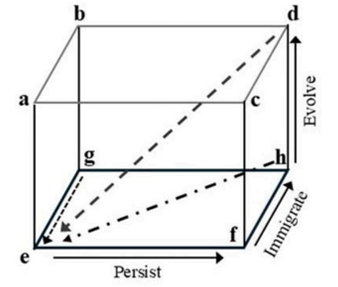
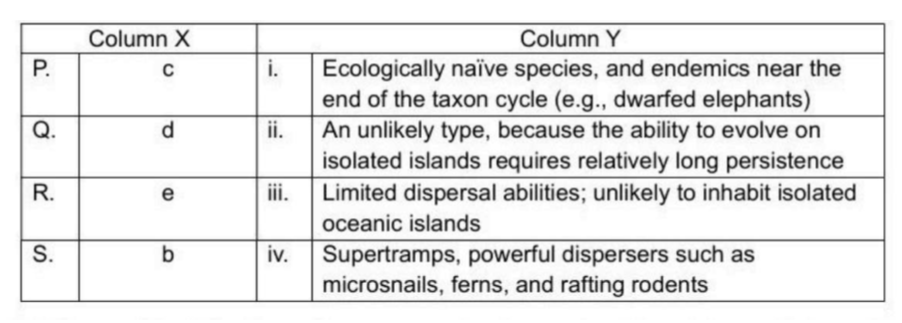
Correct option is C
Correct Answer: (3) less than the sum of the van Der Waals radii.
Explanation:
In hydrogen bonding, the donor (D-H) and acceptor (A) atoms are typically closer than the sum of their van der Waals radii.
The D…A distance in hydrogen bonds is generally less than the sum of the van Der Waals radii, due to the attractive nature of the hydrogen bond pulling the atoms closer.
Van der Waals radii represent the closest distance at which two non-bonded atoms can approach each other. In a hydrogen bond, the D-H and A atoms are brought closer due to the interaction between the partially positive hydrogen atom and the lone pair on the acceptor atom.
Information Booster:
A hydrogen bond occurs when a hydrogen atom covalently bonded to an electronegative atom, like oxygen, nitrogen, or fluorine, interacts with another electronegative atom possessing a lone pair.
The strength of hydrogen bonding depends on the electronegativity of the atoms involved, the distance between them, and the angle of the bond.
The D-H…A distance in hydrogen bonds is typically around 1.5-2.5 Å, which is shorter than the sum of the van Der Waals radii.
Hydrogen bonding plays a crucial role in the properties of water and biological macromolecules such as DNA and proteins.
In water, hydrogen bonds are responsible for its high boiling point, high surface tension, and its ability to dissolve many substances.
Hydrogen bonds are directional, meaning that the strength of the interaction is influenced by the angle between the D-H and A atoms.
Additional Information:
Option 1: one-and-a-half times the sum of the van Der Waals radii. This statement is incorrect because the D…A distance in a hydrogen bond is generally closer than one-and-a-half times the sum of van Der Waals radii.
Option 2: equal to the sum of the van Der Waals radii. This is incorrect because the D…A distance is typically shorter than the sum of the van Der Waals radii due to the nature of the hydrogen bond.
Option 4: twice the sum of the van Der Waals radii. This is incorrect as the hydrogen bond distance is significantly shorter than twice the van Der Waals radii.