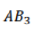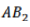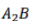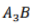Correct option is A
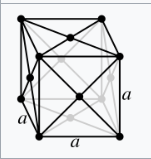
A face-centred unit cell (F) has lattice points at its corners and also at the centres of its six faces.
The face-centered cubic lattice (cF) has lattice points on the faces of the cube, that each gives exactly one half contribution, in addition to the corner lattice points, giving a total of four lattice points per unit cell (1⁄8 × 8 from the corners plus 1⁄2 × 6 from the faces).