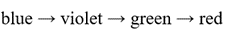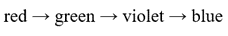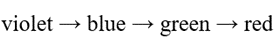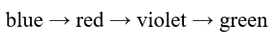Correct option is C
A semiconductor is a material that is between the conductor and insulator in ability to conduct electrical current. Some examples of semiconductors are germanium, silicon, etc. In solids, energy bands are defined as the number of atoms that are in close proximity to one another and the number of electrons that interact with one another. Valence band is essentially described as the energy band made up of valence electrons found in an atomic structure's outermost shell. When given enough energy, these valence electrons transform into free electrons and travel to the conduction band, which results in conductivity. The band with conduction electrons and the lowest occupied energy is known as the conduction band. The energy gap in a crystalline solid is the difference in energy between the electrons' conduction band bottom and valence band. The energy gap in semiconductors is relatively small.
Fluorescence is one of two kinds of emission of light by a substance that has absorbed light or other electromagnetic radiation. Fluorescence occurs when a photon of the incoming radiation is absorbed by an orbital electron in a molecule of the material, exciting it to a higher energy level. The electron then returns to its former energy level by losing energy, emitting another photon of a different frequency, which is the fluorescent glow.

Lower the energy gap for the excitation of the electron, higher will be the wavelength.
VIBGYOR is an acronym that represents the colors of the visible spectrum. The visible spectrum is the range of electromagnetic radiation that is visible to the human eye. It includes all the colors of the rainbow and is typically divided into seven main colors: red, orange, yellow, green, blue, indigo, and violet.
The wavelengths of the colors of the visible spectrum, in nanometers (nm), are as follows:
1. Red: 630-700 nm
2. Orange: 590-620 nm
3. Yellow: 570-590 nm
4. Green: 495-570 nm
5. Blue: 450-495 nm
6. Indigo: 420-450 nm
7. Violet: 380-420 nm