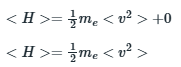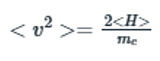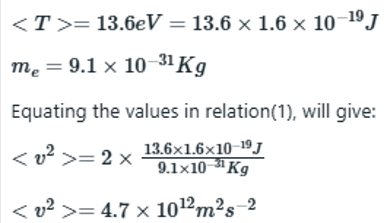Correct option is C
The expectation value
If the system is in a state described by the wave function
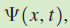
, and the value of the observable a is measured once on each of many identically prepared systems, the average value (also called the expectation value) of all of these measurements is given by
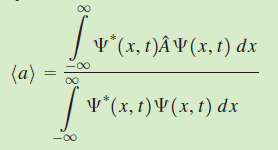
For the case in which
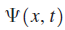
is normalized, the denominator in this expression has the value 1.
Expectation value of energy of an electron is the sum of expectation value of kinetic and potential energy.
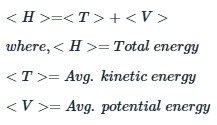
The expectation value of kinetic energy of electron is related to mass of electron and the velocity as:
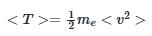
According to the question, the potential energy of the electron inside the well is 0.