Correct option is D
Introduction:
- Arsenic compounds can be broadly categorized as inorganic and organic. Inorganic arsenic compounds are generally more toxic than organic arsenic compounds.
- Within inorganic arsenic, the trivalent form (like in arsenous acid) is typically considered more toxic than the pentavalent form (like in arsenic acid).
- Organic arsenic compounds, such as arsenocholine and arsenobetaine, are generally considered to be of much lower toxicity and are often readily excreted by the body.
- The order of increasing LD50 (from most toxic to least toxic) for the given compounds is:
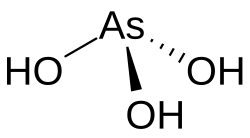
- A. Arsenous Acid (H₃AsO₃) - Trivalent inorganic arsenic, generally most toxic.
- B. Arsenic Acid (H₃AsO₄) - Pentavalent inorganic arsenic, less toxic than the trivalent form.
- D. Dimethylarsinic Acid ((CH₃)₂AsO(OH)) - An organic arsenic compound, significantly less toxic than inorganic forms.
- C. Arsenocholine ((CH₃)₃As⁺CH₂CH₂OH) - An organic arsenic compound, with low toxicity.
- E. Arsenobetaine ((CH₃)₃As⁺CH₂COO) - A very low toxicity organic arsenic compound.
- Thus, the correct arrangement in increasing order of LD50 is:
- Arsenous Acid < Arsenic Acid < Dimethylarsinic Acid < Arsenocholine < Arsenobetaine
- A. Arsenous Acid (H₃AsO₃) < B. Arsenic Acid (H₃AsO₄) < D. Dimethylarsinic Acid ((CH₃)₂AsO(OH)) < C. Arsenocholine ((CH₃)₃As⁺CH₂CH₂OH) < E. Arsenobetaine ((CH₃)₃As⁺CH₂COO).
- The toxicity of arsenic compounds is influenced by their chemical form. Inorganic arsenic is more toxic than organic arsenic.
- Trivalent inorganic arsenic is generally more toxic than pentavalent inorganic arsenic.
- Organic arsenic compounds like arsenocholine and arsenobetaine are typically well-tolerated and rapidly excreted, resulting in much higher LD50 values (lower toxicity) compared to inorganic forms.









 English
English 10 Questions
10 Questions 12 Mins
12 Mins



