Correct option is B
Left to equilibration, many compositions will form a uniform single phase, but depending on the temperature and pressure even a single substance may separate into two or more distinct phases. Within each phase, the properties are uniform but between the two phases properties differ.
The degree of freedom or variance of the system is the minimum number of independent variables such as temperature, pressure and concentration, that must be ascertained so that a given system in equilibrium is completely defined.
For an illustration, we cite an example of water system. If only one phase is present (solid, liquid or gas), then we have to describe values of at least two parameters, namely, temperature and pressure, in order to define the system. For example, we can have liquid water under different conditions of temperature and pressure. Thus, the water system has two degrees of freedom or it is bivariant. Now consider two phases in equilibrium, for example, solid and liquid water, i.e.
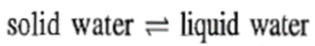
It is well known that the temperature at which a solid melt depends upon the external pressure. Thus, if the value of one of the variables is fixed, the other will automatically have a definite value. The usual melting point of 0 °C is referred to one atm external pressure. In order to describe the above equilibrium completely, we need to specify only one variable-either temperature or pressure; hence the system has one degree of freedom, i.e. it is a univariant system.
Alternatively, the degree of freedom of the system may be defined as the number of factors, such as temperature, pressure and concentration, which can be varied independently without altering the number of phases. For a single-phase system, both temperature and pressure can be varied independently of each other and thus the system is bivariant. For two phases in equilibrium, only one variable can be varied as the other one will automatically have a fixed value and thus the system is univariant.
Consider now the situation at the triple point of water where all the three phases are in equilibrium with each other. This equilibrium is achieved at a temperature of 0.009 8 °C and a pressure of 4.58 mmHg. Any change in the values of these variables, converts the three-phase system into either two-phase or one-phase system. Thus, none of the variables can be changed so long as we want to have all the three phases in equilibrium. Thus, the system has zero degree of freedom, or it is nonvariant.
Phase rule is a general rule which is applicable to all types of reactive and nonreactive systems. In a nonreactive system, we simply have the distribution of various components in different phases without any complications such as the chemical reaction between the components. We derive the rule, first, for a nonreactive system and then show how the same rule can be used for a reactive system.
Consider a heterogeneous system of P phases at equilibrium containing in all C components. Let us start with the assumption that all the C components are present in all P phases. The system at equilibrium can be completely described if we know the values of the variables listed in Table.

Values of these variables can be obtained by solving the equations which are applicable when the system is at equilibrium. There are two types of equations which are available (Table).
Mathematically, we know that the number of variables that can be obtained from a set of equations is equal to the number of equations. Thus if there are as many equations as there are variables, then the temperature, pressure and composition of the whole system in equilibrium can be determined. Such a system, is the nonvariant system. If the number of variables exceeds the number of equations by one, then the equilibrium of the system cannot be determined until one of the variables is arbitrarily chosen. Such a system is called monovariant (or univariant) and has one degree of freedom. Thus we see that the excess of variables over equations, which is called the variance F of the system, is given as











