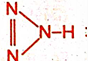
Correct option is A
The correct answer is: (a) Metallic cation
Explanation: Electronic configuration:
1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁹
This corresponds to
Copper (Cu, Z = 29) in
+1 oxidation state (Cu⁺):
• Neutral Cu: [Ar] 3d¹⁰ 4s¹ • Cu⁺: [Ar] 3d¹⁰ • Cu²⁺: [Ar] 3d⁹ ← This matches the configuration given.
Hence, it's a
metallic cation.
Information Booster: •
Cu²⁺ is a common oxidation state of copper. • Transition metals often lose electrons from the
4s orbital first, then 3d.





 English
English 10 Questions
10 Questions 10 Mins
10 Mins