Correct option is B
The correct answer is:
(b) +3
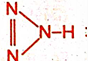
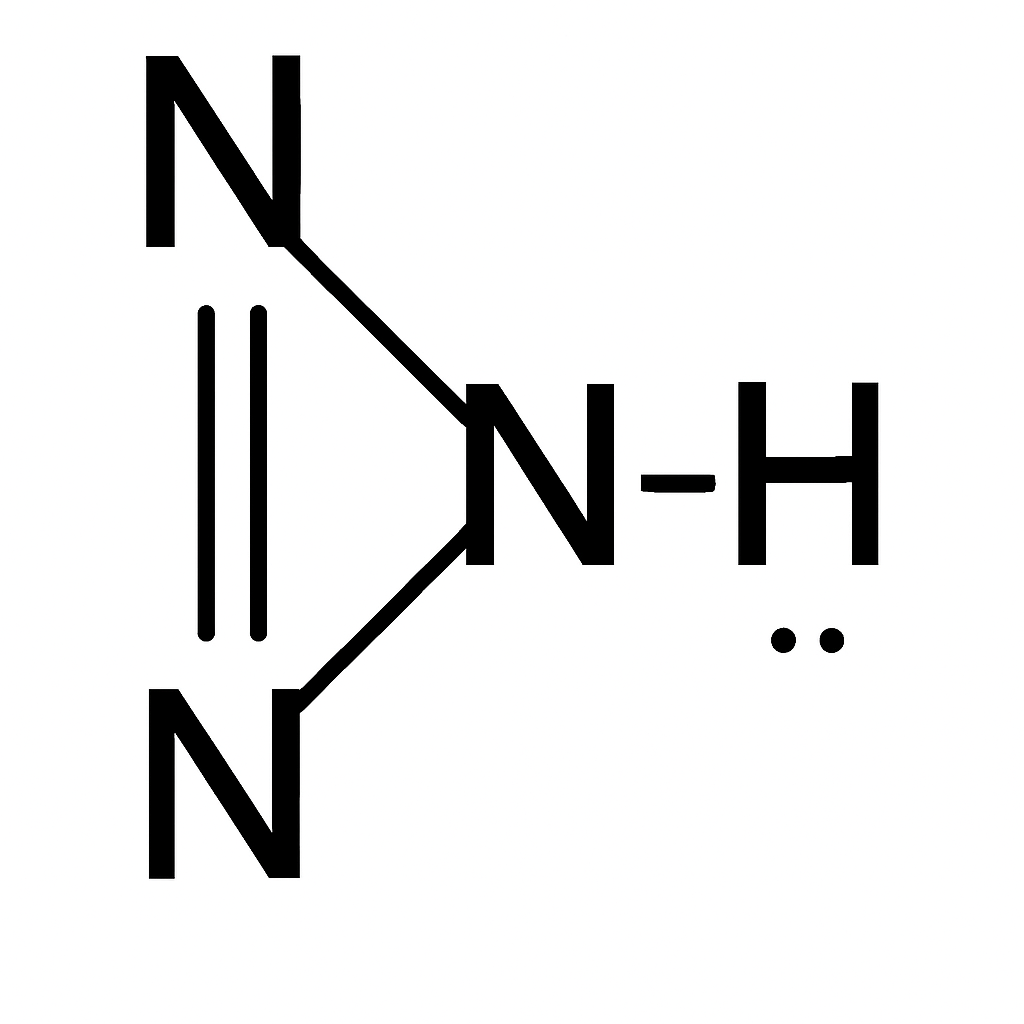
Oxidation Number of Nitrogen with Double Bonds in Hydrazoic Acid (HN₃):
Hydrazoic acid has the molecular formula
HN₃, and one of its resonance structures is:
H–N⁻=N⁺=N
In this structure:
· The
first nitrogen is bonded to hydrogen and another nitrogen: usually has oxidation number
–3.
· The
third nitrogen has a negative formal charge: oxidation number
–1.
· The
central nitrogen is
double bonded to both adjacent nitrogen atoms, and carries a
positive charge.
To calculate the oxidation state of the
central nitrogen:
Let the oxidation numbers be:
· First N (bonded to H) = –3
· Third N = –1
· Central N = x
· Hydrogen = +1
Using the rule that the total oxidation number in a
neutral compound is 0:
(+1) + (–3) + x + (–1) = 0 → x =
+3





 English
English 10 Questions
10 Questions 10 Mins
10 Mins

