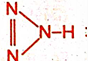
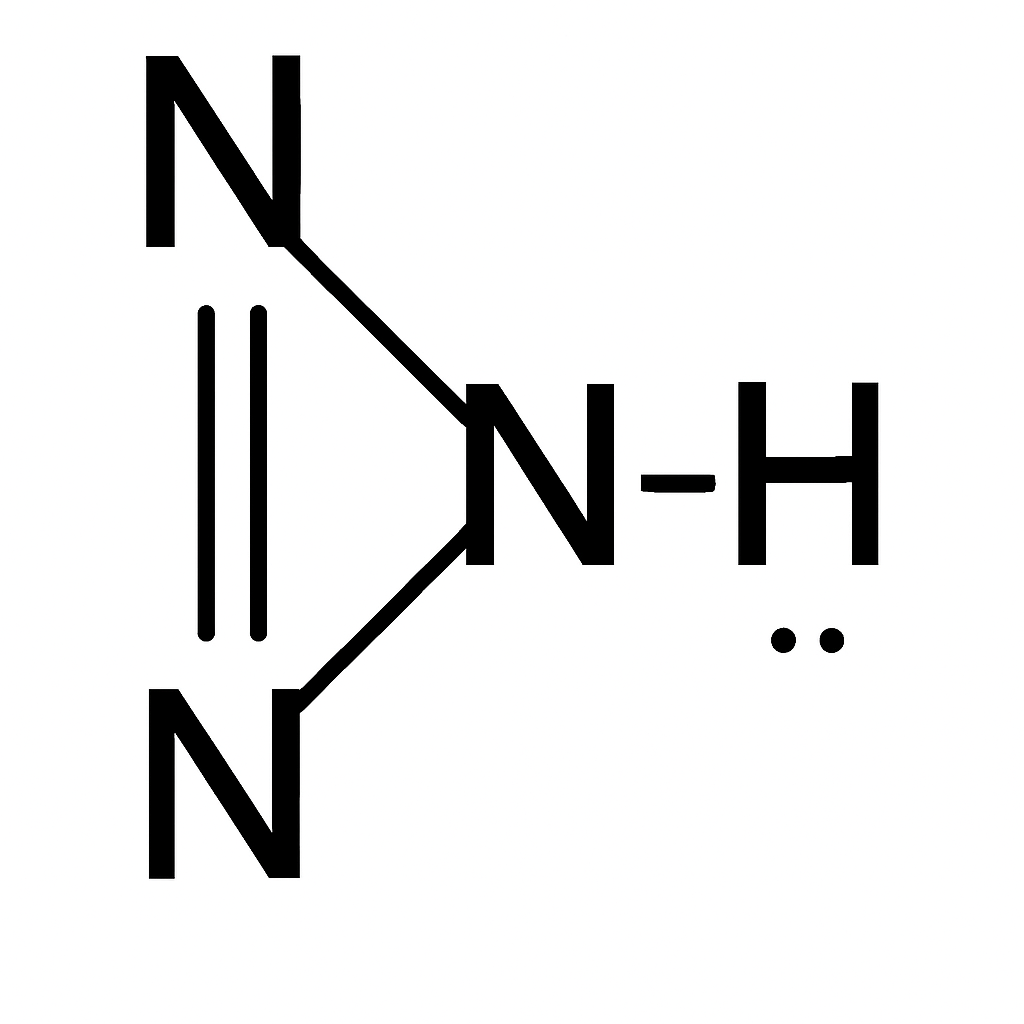
Correct option is A
The correct answer is citric acid
Lemon is widely recognized as a rich source of citric acid, a naturally occurring weak organic acid that is primarily responsible for the fruit’s sour taste. Among all citrus fruits, lemon contains one of the highest concentrations of citric acid, typically ranging between 5% to 8% of its total juice content. The sharp, tangy flavor that characterizes lemon juice is due to this high citric acid content.
Citric acid plays a significant role in plant metabolism, especially in the citric acid cycle (Krebs cycle), and in lemons, it contributes to acidity, preservation, and flavor enhancement. Apart from flavor, citric acid acts as a natural preservative and helps maintain the pH balance of foods. It also prevents oxidation, which protects the color and quality of juices and processed lemon products.
Information Booster:
· Lemons are also a good source of vitamin C (ascorbic acid), but citric acid is the dominant organic acid.
· Citric acid is commercially extracted from lemon juice and used in soft drinks, confectionery, canned foods, jams, and pharmaceuticals.
· Its chemical formula is C₆H₈O₇, and it is valued for its chelating properties, meaning it can bind to metal ions, making it useful in cleaning agents and industrial processes as well.
Additional Knowledge:
· Tartaric acid is commonly found in grapes and tamarind, not in lemons.
· Malic acid is mainly found in apples, pears, and cherries, giving them a milder sourness.
· Acetic acid is the main component of vinegar, and is not naturally abundant in lemons.





 English
English 150 Questions
150 Questions 150 Mins
150 Mins