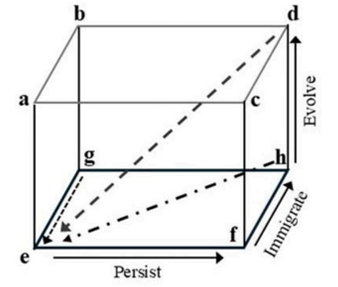
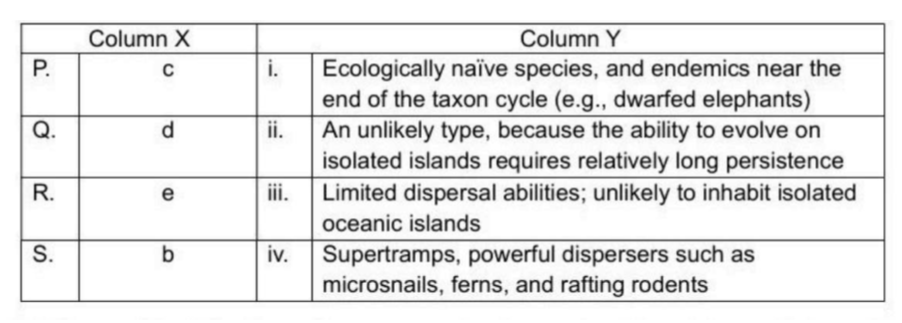
Correct option is A
Explanation-
The molar extinction coefficient (also called molar absorptivity, denoted by ε) is a constant that relates how strongly a chemical species absorbs light at a given wavelength.
It appears in Beer-Lambert Law:
Where:
= Absorbance (unitless)
= Molar extinction coefficient
= Concentration of the solution (mol/L)
= Path length of the cuvette (cm)
Rearranging the equation:
Since:
Absorbance (A) has no units,
Concentration (c) has units mol/L,
Path length (l) has units cm,
Substitute the units into the formula:
= L mol⁻¹ cm⁻¹
L: Liters — unit of volume
mol⁻¹: Per mole — unit of amount of substance
cm⁻¹: Per centimeter — unit of path length
This unit tells you how much light is absorbed per mole of substance in 1 liter solution over a 1 cm path length.
Final answer - option a : L mol⁻¹ cm⁻¹