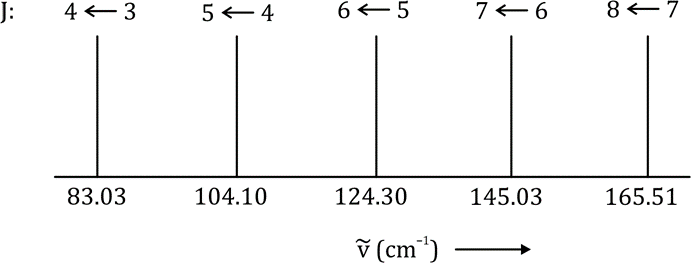
Correct option is C
Rotational (Microwave) spectra of diatomic molecules
Consider a diatomic molecule in which m1 and m2 are the masses of the two atoms and r is the equilibrium bond length, rotating about an axis passing through its centre of gravity.
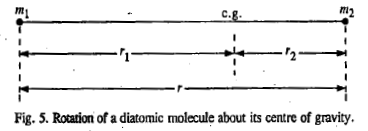
The centre of gravity is defined by the equality of the moments about it
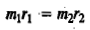
The moment of inertia I of a molecule (rotating as a rigid rotor, not subject to centrifugal forces that tend to distort the molecular geometry and change the moments of inertia ) is defined as
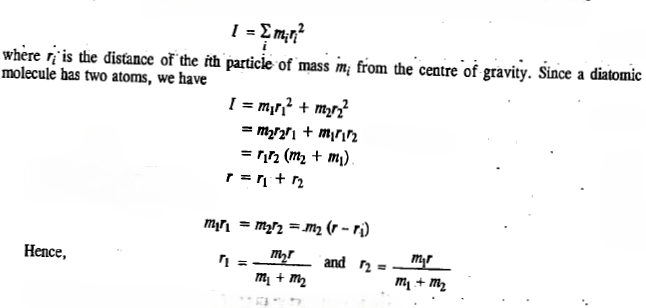
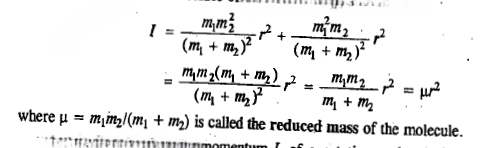


If we want to take into account centrifugal distortion whose effect on the diatomic rotor is to stretch the bond and hence to increase the moment of inertia, and thereby to reduce the rotational constant and hence bring the energy levels closer than in the rigid-rotor approximation, then the energy level expression becomes

Next we need a selection rule to determine the radiative transitions between the rotational energy levels. The derivation of the selection rule is a quantum-mechanical problem; its details need not concern us here. Suffice it to mention that the rotational transitions for a rigid diatomic molecule are governed by the selection rule
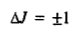
i.e., only those transitions are allowed in which the rotational quantum number changes by unity. The + sign refers to absorption and the - sign to emission of radiation. Microwave-spectra are usually observed as absorption spectra so that the operative part of the selection rule is Δ J = + 1. For a transition taking place from J to J+ 1, the rotational frequency is given by
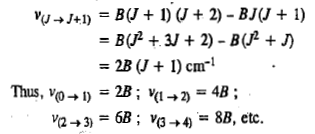
We see that the rotational spectrum of a rigid diatomic molecule consists of a series of lines at 2B, 4B, 6B, 8B etc. Evidently, these lines are equally spaced by an amount of 2B called frequency separation.
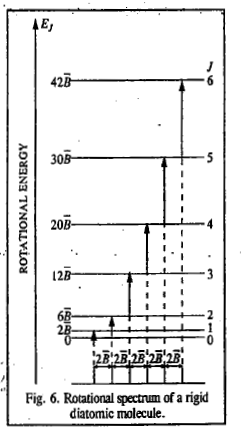
In the given question,
104-83=21
2B=21
B=10.5