Correct option is D
We need to consider how the solvent interacts with the solute. The π electrons of benzene, a soft base, are available for complex formation with the empty orbitals of the cation Ag+, a soft acid. The Ag+ ion is thus solvated favourably by benzene. The species
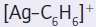
is the complex of the acid Ag+ with π electrons of the weak base benzene.
HSAB is an acronym for "hard and soft (Lewis) acids and bases". HSAB is widely used in chemistry for explaining the stability of compounds, reaction mechanisms and pathways. It assigns the terms 'hard' or 'soft', and 'acid' or 'base' to chemical species. 'Hard' applies to species which are small, have high charge states (the charge criterion applies mainly to acids, to a lesser extent to bases), and are weakly polarizable. 'Soft' applies to species which are big, have low charge states and are strongly polarizable. Essentially, the theory states that soft acids prefer to form bonds with soft bases, whereas hard acids prefer to form bonds with hard bases, all other factors being equal. It can also be said that hard acids bind strongly to hard bases and soft acids bind strongly to soft bases.
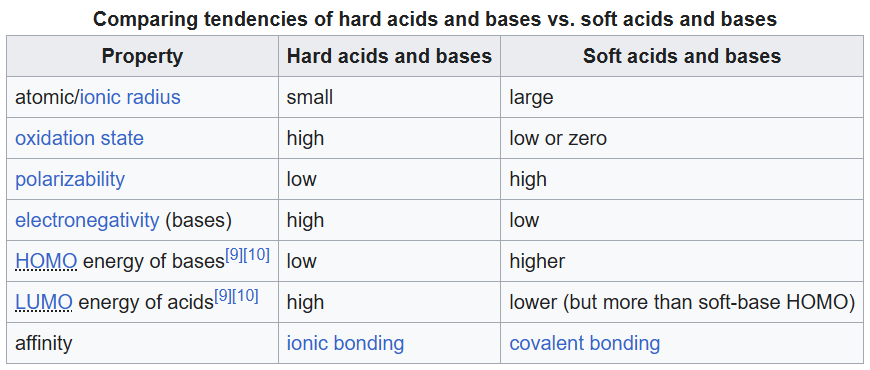
Ag+ is a soft acid and benzene is a soft base.







