Correct option is B
Explanation-
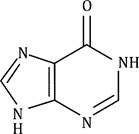
Hypoxanthine is a purine base that is found in inosine, a nucleoside (base + sugar). In DNA, hypoxanthine does not normally occur, but it can appear: As part of inosine in tRNA Or due to deamination of adenine, a process that converts adenine to hypoxanthine.
Inosine monophosphate (IMP) is an intermediate in purine biosynthesis, leading to AMP and GMP.
Base Pairing Rules-
In normal DNA:
Adenine (A) pairs with Thymine (T) — 2 hydrogen bonds.
Guanine (G) pairs with Cytosine (C) — 3 hydrogen bonds.
Hypoxanthine (in inosine) - Structurally similar to guanine, but lacks the amino group at position 2.
The nitrogenous bases with pairing Strength with Hypoxanthine (I) and their reasons-
Cytosine (C) - Strongest - 2 stable hydrogen bonds — good shape and polarity match.
Adenine (A) - Moderate - Forms 2 hydrogen bonds, less stable than with cytosine.
Thymine (T) - Weaker - One or two weak hydrogen bonds possible.
Guanine (G) - Weakest or negligible - Bad shape match — both are purines; sterically unfavorable.
So, the correct answer is option b - Cytosine > Adenine > Thymine > Guanine.