Correct option is A
The correct answer is option ( a ) 12, 3, 15
Given : Benzene molecule
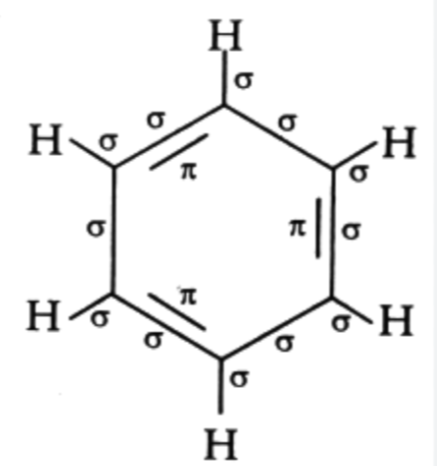
- Benzene is a hexagonal ring structure with 6 carbon atoms and 6 hydrogen atoms.
- Each carbon is bonded to:
- One hydrogen atom via a σ-bond.
- Two neighboring carbon atoms via one σ-bond each.
- Alternating double bonds form π-bonds.
Count the σ-bonds :
- C - H σ-bonds : 6
- C - C σ-bonds : 6
Total σ-bonds=6(C-H)+6(C-C)=12\text{Total } \sigma \text{-bonds} = 6 \, (\text{C-H}) + 6 \, (\text{C-C}) = 12Total σ-bonds=6(C-H)+6(C-C)=12
Count the π-bonds :
- The alternating double bonds in benzene consist of 3 π-bonds, one for each double bond.
Total π-bonds=3\text{Total } \pi \text{-bonds} = 3Total π-bonds=3
Count the total bonds :
Total bonds=σ-bonds+π-bonds=12+3=15\text{Total bonds} = \text{σ-bonds} + \text{π-bonds} = 12 + 3 = 15 Total bonds=σ-bonds+π-bonds=12+3=15
σ-bonds: 121212
π-bonds: 333
Total bonds: 151515





 English
English 100 Questions
100 Questions 90 Mins
90 Mins


 at 298 K will produce 47% of
at 298 K will produce 47% of





