Correct option is B
The correct critical temperature Tc derived from the van der Waals equation is:
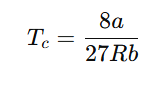
Explanation: The van der Waals equation of state for real gases is:
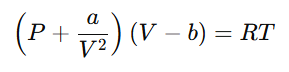
Critical constants are obtained by solving the following conditions at the critical point:
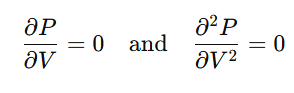
Information Booster:
1. The van der Waals equation extends the ideal gas law to include the non-zero size of gas molecules and the interactions between them (both of which depend on the specific substance).
2. Critical temperature (Tc) is the temperature above which a gas cannot be liquefied regardless of pressure.
3. R is the universal gas constant (8.314 JK-1mol-1)
4. aa and b bare experimentally determinable, substance-specific constants. The constant aa expresses the strength of the molecular interactions. The constant bb denotes an excluded molar volume.





 English
English 150 Questions
150 Questions 150 Mins
150 Mins

