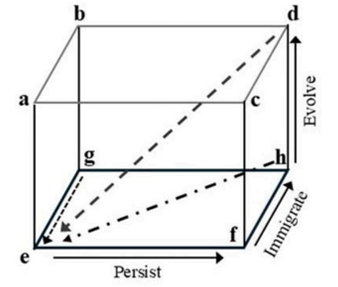
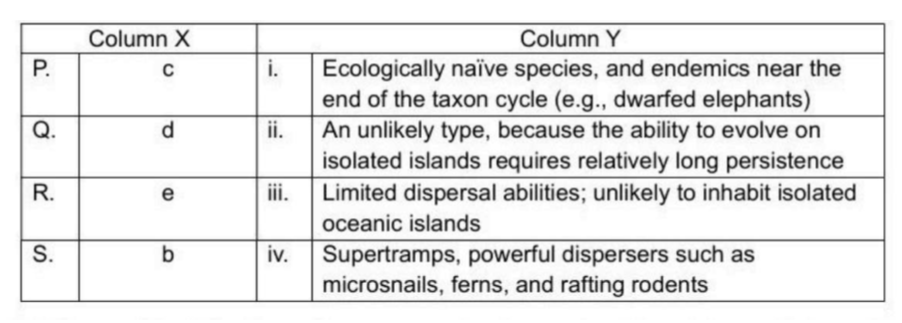
Correct option is A
The energy required to break a single covalent bond varies depending on the type of bond and the atoms involved. Typically, for covalent bonds, the bond dissociation energy (the energy required to break the bond and separate the atoms) falls within the range of 200-500 kJ/mol. This is the standard energy range for common single covalent bonds between atoms in molecules like hydrogen, oxygen, or carbon compounds.
Information Booster:
The bond dissociation energy for single covalent bonds typically falls between 200 and 500 kJ/mol.
Covalent bonds involve the sharing of electrons between two atoms, and breaking them requires overcoming the attractive forces holding the atoms together.
The energy required depends on factors such as the size and electronegativity of the atoms involved in the bond.
For example, the bond dissociation energy for the H-H bond is approximately 435 kJ/mol, which is within this range.
The exact energy can vary slightly depending on the specific bond, such as in hydrocarbons or halogen compounds.
Stronger bonds, like triple bonds, require more energy to break compared to single bonds.
Additional Knowledge:
80-150 kJ/mol (option 2) is too low for a single covalent bond as it usually corresponds to weaker forces like van der Waals interactions or hydrogen bonds, not covalent bonds.
600-900 kJ/mol (option 3) represents the energy range for breaking stronger bonds, such as triple bonds (e.g., in nitrogen molecules), not typical single covalent bonds.
20-70 kJ/mol (option 4) represents energies that are much too low and would be more relevant for very weak intermolecular forces, not covalent bonds.